
Concept explainers
(a)
Interpretation:
Whether the given substrate undergoes E2 elimination step with base
Concept introduction:
Answer to Problem 7.29P
The given substrate cannot undergo E2 elimination step with base
Explanation of Solution
The given substrate is
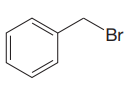
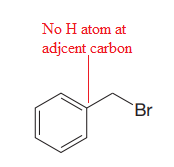
Thus, the given substrate cannot undergo
No H atom on adjacent carbon atom.
(b)
Interpretation:
Whether the given substrate undergoes E2 elimination step with base
Concept introduction:
Answer to Problem 7.29P
The given substrate undergoes E2 elimination step.
The E2 elimination step for the given substrate is drawn as:
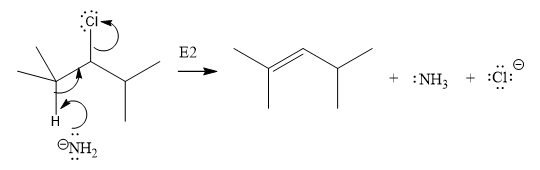
Explanation of Solution
The given substrate is:
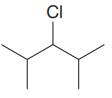
In the above given substrate, Cl acts as a leaving group with one H atom on each side. Any one of the hydrogen atom is taken away forming a C=C bond. The first curved arrow is drawn from the lone pair of nitrogen atom of a base

The product of
(c)
Interpretation:
Whether the given substrate undergo E2 elimination step with base
Concept introduction:
Answer to Problem 7.29P
The given substrate undergoes E2 elimination step.
The E2 elimination step for the given substrate is drawn as:
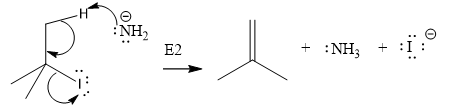
Explanation of Solution
The given substrate is:
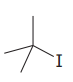
In the above given substrate,
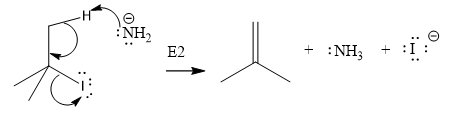
The product of
(d)
Interpretation:
Whether the given substrate undergoes E2 elimination step with base
Concept introduction:
Answer to Problem 7.29P
The given substrate cannot undergoes E2 elimination step with base
Explanation of Solution
The given substrate is
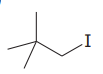
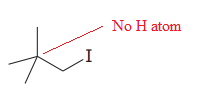
Thus, the given substrate cannot undergo
There is no H atom on the adjacent carbon atom.
(e)
Interpretation:
Whether the given substrate undergo E2 elimination step with base
Concept introduction:
The
Answer to Problem 7.29P
The given substrate undergoes E2 elimination step.
The E2 elimination step for the given substrate is drawn as:
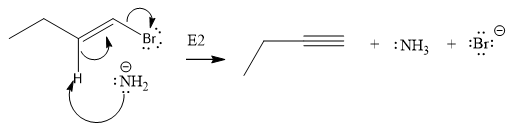
Explanation of Solution
The given substrate is
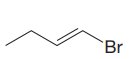
In the above given substrate,
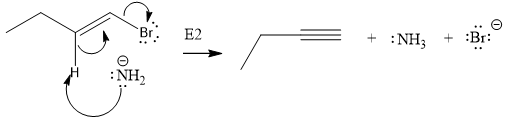
The product of
Want to see more full solutions like this?
Chapter 7 Solutions
Get Ready for Organic Chemistry
- Drawing the Product(s) of a Mechanistic Step. For the reactions that have TWO starting materials, indicate/label which species is the nucleophile and which isthe electrophile. Then, by following the curved electron-pushing arrows, draw the product(s) of the following reaction steps. Finally, identify and label what type of elementary step is represented for each mechanism.arrow_forwardWhich of the following would proceed through a carbocation rearrangement E1 reaction?, biingarrow_forwardDraw the reaction mechanism (with arrows) of the following reaction?arrow_forward
- If the anionic species shown were to eliminate a leaving group, the three possi- bilities would be H3C¯, CI, or CH30¯. Draw the curved arrow notation and the products for each of these elimination steps. Which is the major product? Why? CIarrow_forwardWhich reaction would not be favorable?arrow_forwardFrom the following reactions couples Which of them will go faster ? Explainarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





