
(a)
Interpretation:
The curved arrow notations showing all possible
Concept introduction:
Curved arrows are used to represent the movement of electrons in a reaction mechanism. The arrow starts on an electron-rich atom or an electron-rich region such as a pi bond. It ends on an electron poor atom when the movement results in the formation of a new sigma bond. If the result is the formation of a pi bond, the arrow ends in the region between the two atoms that form the bond.
A carbocation is a positively charged carbon atom that is electron-poor, two electrons short of an octet. It is unstable because it is a charged species.
A
A
The numbering in the shift label simply signifies that a hydride or a methyl group migrates from one carbon to an adjacent one. The numbering is not related to the root chain atom numbering.
Answer to Problem 7.36P
The possible
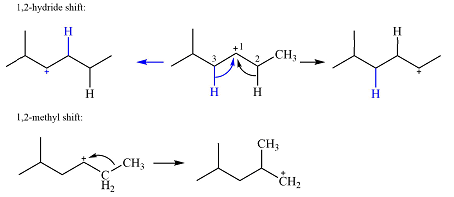
Explanation of Solution
The structure of the carbocation is
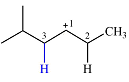
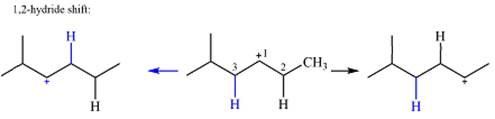
The charge is on the carbon numbered 1. There are two hydrogen atoms on the adjacent carbons C2 and C3. These are the ones that can shift to C1 in the two possible
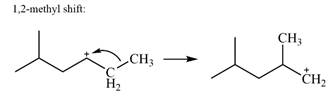
There is only one methyl group on the carbon adjacent to C1, attached to C2. Shifting of this methyl to C1 results in shifting of the charge to C2. Therefore, the
A hydride (
(b)
Interpretation:
The curved arrow notations showing all possible
Concept introduction:
Curved arrows are used to represent the movement of electrons in a reaction mechanism. The arrow starts on an electron-rich atom or an electron-rich region such as a pi bond. It ends on an electron poor atom when the movement results in the formation of a new sigma bond. If the result is the formation of a pi bond, the arrow ends in the region between the two atoms that form the bond.
A carbocation is a positively charged carbon atom that is electron-poor, two electrons short of an octet. It is unstable because it is a charged species.
A
A
The numbering in the shift label simply signifies that a hydride or a methyl group migrates from one carbon to an adjacent one. The numbering is not related to the root chain atom numbering.
Answer to Problem 7.36P
The possible
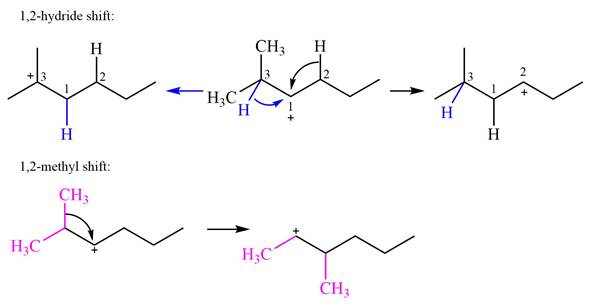
Explanation of Solution
The structure of the given carbocation is
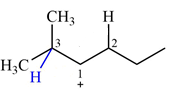
There are two hydrogen atoms on adjacent carbons C2 and C3 that can shift to the positively charged carbon C1 in two possible
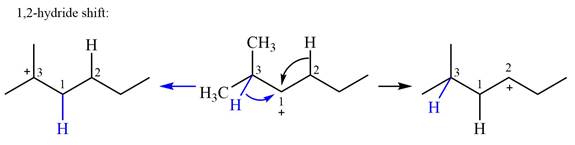
There are two methyl groups attached to a carbon adjacent to C1. Both are on the same carbon C3, therefore, shifting of either one will give the same product.
Therefore, the
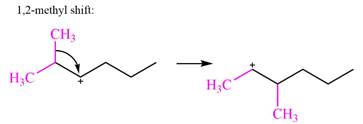
A hydride (
(c)
Interpretation:
The curved arrow notations showing all possible
Concept introduction:
Curved arrows are used to represent the movement of electrons in a reaction mechanism. The arrow starts on an electron-rich atom or an electron-rich region such as a pi bond. It ends on an electron poor atom when the movement results in the formation of a new sigma bond. If the result is the formation of a pi bond, the arrow ends in the region between the two atoms that form the bond.
A carbocation is a positively charged carbon atom that is electron-poor, two electrons short of an octet. It is unstable because it is a charged species.
A
A
The numbering in the shift label simply signifies that a hydride or a methyl group migrates from one carbon to an adjacent one. The numbering is not related to the root chain atom numbering.
Answer to Problem 7.36P
The possible
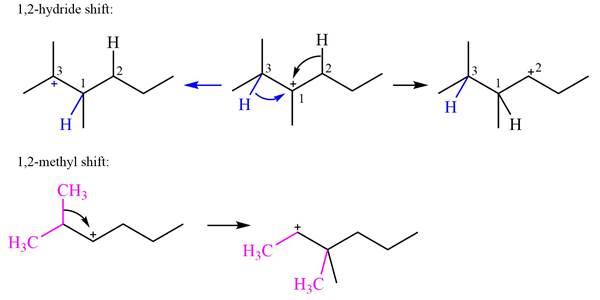
Explanation of Solution
The structure of the given carbocation is
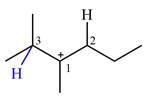
There are two hydrogen atoms on adjacent carbons C2 and C3 that can shift to the positively charged carbon C1 in two possible
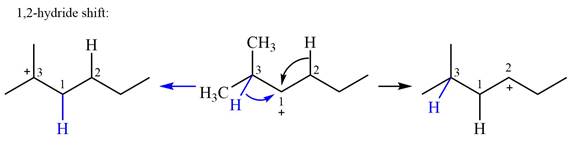
There are two methyl groups attached to a carbon adjacent to C1. Both are on the same carbon C3; therefore, shifting of either one will give the same product.
Therefore, the
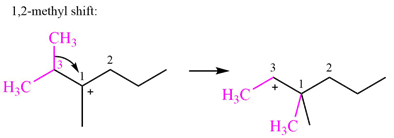
A hydride (
(d)
Interpretation:
The curved arrow notations showing all possible
Concept introduction:
Curved arrows are used to represent the movement of electrons in a reaction mechanism. The arrow starts on an electron-rich atom or an electron-rich region such as a pi bond. It ends on an electron poor atom when the movement results in the formation of a new sigma bond. If the result is the formation of a pi bond, the arrow ends in the region between the two atoms that form the bond.
A carbocation is a positively charged carbon atom that is electron-poor, two electrons short of an octet. It is unstable because it is a charged species.
A
A
The numbering in the shift label simply signifies that a hydride or a methyl group migrates from one carbon to an adjacent one. The numbering is not related to the root chain atom numbering.
Answer to Problem 7.36P
The possible
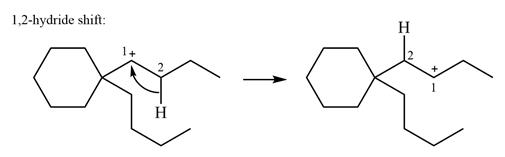
As there are no methyl groups on the carbon adjacent to the charge bearing carbon C1, a
Explanation of Solution
The structure of the given carbocation is
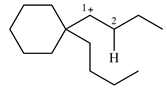
There is only one hydrogen atom on an adjacent carbon, C2, that can shift to the positively charged carbon C1 in a possible
Therefore, the
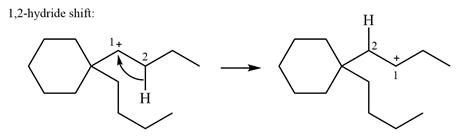
There are no methyl groups attached to the carbon adjacent to C1. Therefore, a
A hydride (
(e)
Interpretation:
The curved arrow notations showing all possible
Concept introduction:
Curved arrows are used to represent the movement of electrons in a reaction mechanism. The arrow starts on an electron-rich atom or an electron-rich region such as a pi bond. It ends on an electron poor atom when the movement results in the formation of a new sigma bond. If the result is the formation of a pi bond, the arrow ends in the region between the two atoms that form the bond.
A carbocation is a positively charged carbon atom that is electron-poor, two electrons short of an octet. It is unstable because it is a charged species.
A
A
The numbering in the shift label simply signifies that a hydride or a methyl group migrates from one carbon to an adjacent one. The numbering is not related to the root chain atom numbering.
Answer to Problem 7.36P
The possible
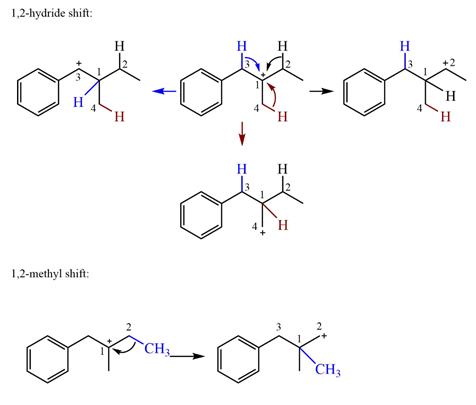
Explanation of Solution
The structure of the given carbocation is
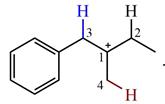
There are three hydrogen atoms on carbon atoms adjacent to the charge carrying carbon. They are on C2, C3, and C4.
Shifting of the hydride on C2 results in the charge shifting to C2, as shown in the product on the right.
Shifting of the hydride on C3 results in the charge shifting to C3, as shown in the product on the left.
Shifting of the hydride on C4 results in the charge shifting to C4, as shown in the product below the given carbocation.
Therefore, the
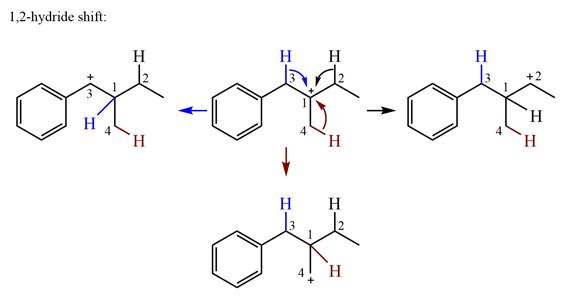
There is one methyl group attached to the carbon adjacent to C1. Shifting of the methyl group on C2 to C1 results in C1 becoming a tertiary carbon and the charge shifting to C2.
Therefore, the possible
A hydride (
(f)
Interpretation:
The curved arrow notations showing all possible
Concept introduction:
Curved arrows are used to represent the movement of electrons in a reaction mechanism. The arrow starts on an electron-rich atom or an electron-rich region such as a pi bond. It ends on an electron poor atom when the movement results in the formation of a new sigma bond. If the result is the formation of a pi bond, the arrow ends in the region between the two atoms that form the bond.
A carbocation is a positively charged carbon atom that is electron-poor, two electrons short of an octet. It is unstable because it is a charged species.
A
A
The numbering in the shift label simply signifies that a hydride or a methyl group migrates from one carbon to an adjacent one. The numbering is not related to the root chain atom numbering.
Answer to Problem 7.36P
The possible
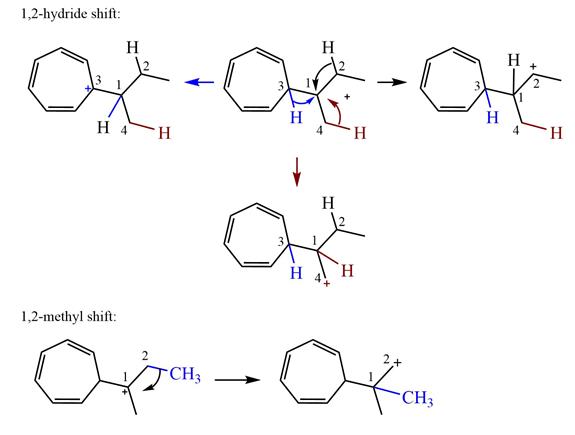
Explanation of Solution
The structure of the given carbocation is
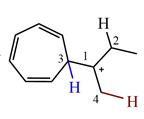
There are three hydrogen atoms on carbon atoms adjacent to the charge carrying carbon. They are on C2, C3, and C4.
Shifting of the hydride on C2 results in the charge shifting to C2, as shown in the product on the right.
Shifting of the hydride on C3 results in the charge shifting to C3, as shown in the product on the left.
Shifting of the hydride on C4 results in the charge shifting to C4, as shown in the product below the given carbocation.
Therefore, the
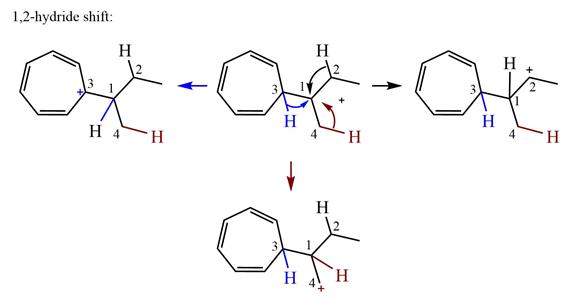
There is one methyl group attached to a carbon adjacent to C1. Shifting of the methyl group on C2 to C1 results in C1 becoming a tertiary carbon and the charge shifting to C2.
Therefore, the possible
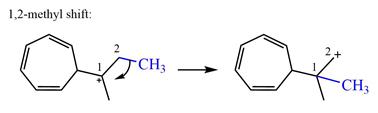
A hydride (
Want to see more full solutions like this?
Chapter 7 Solutions
EBK GET READY FOR ORGANIC CHEMISTRY
- Consider the reaction between 2-methyl-2-butanol and HBr, shown below. но HBr Brarrow_forwardDraw the missing curved arrow notation to incidate how the carbocation in left box rearranges to the carbocation in the right box 1st attempt See Periodic Ta Add the missing curved arrow notation. 3 OF 25 QUESTIONS COMPLETEDarrow_forward3. Draw the major product in the following reaction and reaction mechanism. Remember when drawing a reaction mechanism, you need to show all nonbonding electron pairs, formal charges, electron flow arrows, and all resonance structures. What is the name of this mechanism? Determine and circle whether the substituent is an activator or deactivator. Explain what is meant by being an activator or deactivator and circle the single resonance structure that supports your decision regarding the substituent in the starting material. OH CH₂CH₂CH₂Br FeBrarrow_forward
- Solve correctly please, with some explanation also. Draw the product(s) for the following radical reaction where one bond breaks and another bond is formed. The curved arrows indicate the movement of an unpaired electron.arrow_forward3. Will the following carbocations rearrang e? If so, draw the rearrangement using a curved arrow and the new carbocation. You may have to draw in the H-atom and its bond to properly illustrate the hydride shift. Two will not rearrange.arrow_forwardComplete the reaction in the drawing area below by adding the major products to the right-hand side. If there won't be any products, because nothing will happen under these reaction conditions, check the box under the drawing area instead. Note: if the products contain one or more pairs of enantiomers, don't worry about drawing each enantiomer with dash and wedge bonds. Just draw one molecule to represent each pair of enantiomers, using line bonds at the chiral center. More... i x + P C 1. NaOMe Click and drag to start drawing a structure. 2. Harrow_forward
- Please help with these and include the steps in the reaction so i can better understand how they work! just 1 or two of them is finearrow_forwardIn (e), please note that the heterocyclic oxygen atoms contains two lone pairs that could contribute to the resonance stabilization of any proposed intermediate.arrow_forwardTertiary alkyl halides readily undergo elimination via an E1 mechanism when they react with a weak base, as shown in the video below. Consider the reaction of (2R,3R)-2-bromo-2,3-dimethyl-1-phenylbutane with methanol. CH3OH 60°Carrow_forward
- refer to image, please explain in detail, showing all steps. will rate!arrow_forwardEdit the reaction by drawing all steps in the appropriate boxes and connecting them with reaction arrows. Add charges where needed. Electron flow arrows should start on an atom or a bond and should end on an atom, bond, or location where a new bond should be created.arrow_forwardConsider the molecule given below. In theory, there are only two inequivalent hydrogens in this molecule that could be substituted by Br in a free radical bromination – circle them. Put an asterisk to mark the one most likely to be substituted first. However, there are 5 possible products from free radical bromination. Draw all the products and show using arrow formalism how the intermediate radicals leading to these products formedarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
