
(a)
Interpretation: The mechanism of given nucleophilic substitution reaction is to be determined and the products, along with their stereochemistry, are to be drawn.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any
Answer to Problem 7.63P
The mechanism of given nucleophilic substitution reaction is
Explanation of Solution
The structure of the given
In
However, in the given reaction halogen atom is not attached to chiral carbon. It is bonded to non-chiral carbon atom. The attack of nucleophile has not affect on the stereochemistry of reactant as shown in Figure 1.
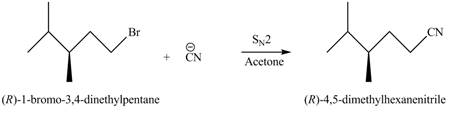
Figure 1
The stereochemistry of reactant and product is same.
The mechanism of given nucleophilic substitution reaction is
(b)
Interpretation: The mechanism of given nucleophilic substitution reaction is to be determined and the products, along with their stereochemistry, are to be drawn.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron rich chemical species that contains negative charge or lone pair of electrons are known as a nucleophile. In a nucleophilic substitution reaction, nucleophile takes the position of leaving group by attacking the electron deficient carbon atom.
Answer to Problem 7.63P
The mechanism of given nucleophilic substitution reaction is
Explanation of Solution
The structure of the given alkyl halide shows that a carbon atom, on which bromine atom is present, is bonded to two other carbon atoms. Hence, the bromine atom is bonded to secondary carbon atom. The removal of bromine atom leads to the formation of secondary carbocation. The secondary carbocation can undergo nucleophilic substitution reaction through both
In
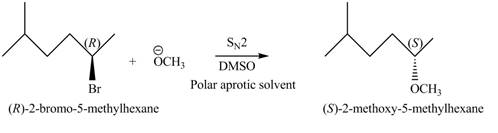
Figure 2
The mechanism of given nucleophilic substitution reaction is
(c)
Interpretation: The mechanism of given nucleophilic substitution reaction is to be determined and the products, along with their stereochemistry, are to be drawn.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron rich chemical species that contains negative charge or lone pair of electrons are known as a nucleophile. In a nucleophilic substitution reaction, nucleophile takes the position of leaving group by attacking the electron deficient carbon atom.
Answer to Problem 7.63P
The mechanism of given nucleophilic substitution reaction is
Explanation of Solution
The structure of the given alkyl halide shows that carbon atom, on which bromine atom is present, is bonded to three other carbon atoms. Hence, the bromine atom is bonded to tertiary carbon atom. The removal of bromine atom leads to the formation of planer tertiary carbocation. The tertiary carbocation is most likely to undergo nucleophilic substitution reaction by
In
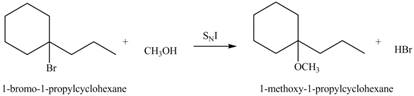
Figure 3
The mechanism of given nucleophilic substitution reaction is
(d)
Interpretation: The mechanism of given nucleophilic substitution reaction is to be determined and the products, along with their stereochemistry, are to be drawn.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron rich chemical species that contains negative charge or lone pair of electrons are known as a nucleophile. In a nucleophilic substitution reaction, nucleophile takes the position of leaving group by attacking the electron deficient carbon atom.
Answer to Problem 7.63P
The mechanism of given nucleophilic substitution reaction is
Explanation of Solution
The structure of the given alkyl halide shows that a carbon atom, on which iodine atom is present, is bonded to two other carbon atoms. Hence, the iodine atom is bonded to secondary carbon atom. The removal of iodine atom leads to the formation of secondary carbocation. The secondary carbocation can undergo nucleophilic substitution reaction through both
In
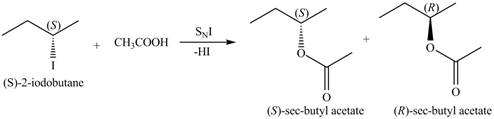
Figure 4
The mechanism of given nucleophilic substitution reaction is
(e)
Interpretation: The mechanism of given nucleophilic substitution reaction is to be determined and the products, along with their stereochemistry, are to be drawn.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron rich chemical species that contains negative charge or lone pair of electrons are known as a nucleophile. In a nucleophilic substitution reaction, nucleophile takes the position of leaving group by attacking the electron deficient carbon atom.
Answer to Problem 7.63P
The mechanism of given nucleophilic substitution reaction is
Explanation of Solution
The structure of the given alkyl halide shows that a carbon atom, on which bromine atom is present, is bonded to two other carbon atoms. Hence, the bromine atom is bonded to secondary carbon atom. The removal of bromine atom leads to the formation of secondary carbocation. The secondary carbocation can undergo nucleophilic substitution reaction through both
In
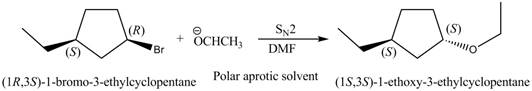
Figure 5
The mechanism of given nucleophilic substitution reaction is
(f)
Interpretation: The mechanism of given nucleophilic substitution reaction is to be determined and the products, along with their stereochemistry, are to be drawn.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron rich chemical species that contains negative charge or lone pair of electrons are known as a nucleophile. In a nucleophilic substitution reaction, nucleophile takes the position of leaving group by attacking the electron deficient carbon atom.
Answer to Problem 7.63P
The mechanism of given nucleophilic substitution reaction is
Explanation of Solution
The structure of the given alkyl halide shows that a carbon atom, on which bromine atom is present, is bonded to two other carbon atoms. Hence, the bromine atom is bonded to secondary carbon atom. The removal of bromine atom leads to the formation of secondary carbocation. The secondary carbocation can undergo nucleophilic substitution reaction through both
In
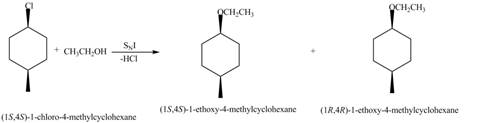
Figure 6
The mechanism of given nucleophilic substitution reaction is
Want to see more full solutions like this?
Chapter 7 Solutions
ORGANIC CHEMISTRY LL W/SSM+CONNECT+KIT
- Draw a stepwise mechanism for the following reaction and explain the stereochemistry of the product.arrow_forwardMuscalure, the sex pheromone of the common housefly, can be preparedby a reaction sequence that uses two nucleophilic substitutions. Identifycompounds A–D in the following synthesis of muscalure.arrow_forwardOne step in the synthesis of the antihistamine fexofenadine involves acid-catalyzed hydration of the triple bond in A. Draw a stepwise mechanism for this reaction and explain why only ketone B is formed.arrow_forward
- Draw the organic product of each reaction (d).arrow_forwardDevise a synthesis of each compound from cyclopentane and any other required organic or inorganic reagents.arrow_forwardDetermine the mechanism of nucleophilic substitution of each reaction and draw the productincluding stereochemistry.arrow_forward
- Draw a stepwise mechanism for the following reduction.arrow_forwardDevise a synthesis of each compound from aniline (C6H5NH2) as starting material.arrow_forwardDevise a synthesis of each compound from benzene. You may also useany organic compounds having four or fewer carbons and any requiredinorganic reagents.arrow_forward
- Devise a synthesis of X from the given starting materials. You may useany organic or inorganic reagents. Account for the stereochemistryobserved in X.arrow_forwardDimethyl cyclopropanes can be prepared by the reaction of an α, β-unsaturated carbonyl compound X with two equivalents of a Wittigreagent Y. Draw a stepwise mechanism for this reaction.arrow_forward1. how to synthesize a and barrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





