
GEN. ORGANIC & BIO CHEM W/ CODE >CUSTO
LATEST Edition
ISBN: 9781323035559
Author: Timberlake
Publisher: Pearson Custom Publishing
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 7, Problem 7.72UTC
Using the models of the molecules
(black = C, white = H, yellow = S, red = 0), determine each of the following for models of compounds I and 2: (7.1, 7.2, 7.3)
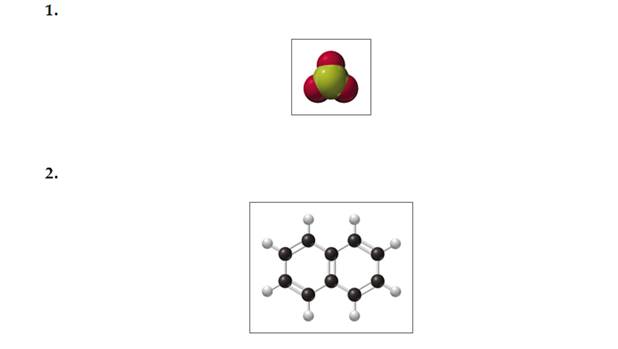
a. molecular formula
b. molar mass
c. number of moles in 10.0 g
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Methane CHis a simple organic molecule that is used as a fuel—it is the major component of natural gas. The bottom of the ocean contains large amounts of methane hydrate (also called methane ice or methane clathrate), enough that it has been suggested as a major source of fuel. The methane hydrate contains 6 moles water for each mole of methane.What percentage of methane hydrate is methane?
blank1 - Numeric Answer %
What percentage is water?
blank2 - Numeric Answer %
The active ingredient of Benadryl© Chesty Forte Cough Liquid is Guaiphenesin an expectorant drug used to assist the expectoration ('bringing up') of phlegm from the airways in acute respiratory tract infections. A 200.0 mL bottle of Benadryl© Chesty Forte Cough contains 4.00 g of Guaiphenesin (C10H14O4).
If the recommended dose for an adult is 350 mg of Guaiphenesin. How many moles of Guaiphenesin does this equal? (answer to 2 significant figures)
A student reacted 3.01g salicylic acid with 5.98mL acetic anhydride and isolated 2.85g Asprin.
A) Calculate moles of salicylic acid used.
B) Calculate the mass of acetic anhydride used.
C) Calculate moles of acetic anhydride used.
D) using moles of limiting reactant present, calculate the theoretical yield of aspirin.
E) Calculate the percent yield of aspirin synthesis.
Chapter 7 Solutions
GEN. ORGANIC & BIO CHEM W/ CODE >CUSTO
Ch. 7.1 - What is a mole?Ch. 7.1 - What is Avogadro’s number?Ch. 7.1 - Calculate each of the following: a. number of C...Ch. 7.1 - Calculate each of the following: a. number of Li...Ch. 7.1 - Calculate each of the following quantities in 200...Ch. 7.1 - Calculate each of the following quantities in...Ch. 7.1 - Prob. 7.7QAPCh. 7.1 - Aluminum sulphate, Al2SO43 , is used in some...Ch. 7.2 - Prob. 7.9QAPCh. 7.2 - Prob. 7.10QAP
Ch. 7.2 - Prob. 7.11QAPCh. 7.2 - Prob. 7.12QAPCh. 7.2 - Prob. 7.13QAPCh. 7.2 - Prob. 7.14QAPCh. 7.2 - Prob. 7.15QAPCh. 7.2 - Prob. 7.16QAPCh. 7.2 - Prob. 7.17QAPCh. 7.2 - Prob. 7.18QAPCh. 7.2 - Prob. 7.19QAPCh. 7.2 - Prob. 7.20QAPCh. 7.2 - Prob. 7.21QAPCh. 7.2 - Prob. 7.22QAPCh. 7.2 - Prob. 7.23QAPCh. 7.2 - Prob. 7.24QAPCh. 7.2 - Prob. 7.25QAPCh. 7.2 - Prob. 7.26QAPCh. 7.3 - Determine whether each of the following chemical...Ch. 7.3 - Determine whether each of the following chemical...Ch. 7.3 - Balance each of the following chemical equations:...Ch. 7.3 - Balance each of the following chemical equations:...Ch. 7.3 - Balance each of the following chemical equations:...Ch. 7.3 - Balance each of the following chemical equations:...Ch. 7.4 - Prob. 7.33QAPCh. 7.4 - Prob. 7.34QAPCh. 7.4 - Prob. 7.35QAPCh. 7.4 - Prob. 7.36QAPCh. 7.4 - Prob. 7.37QAPCh. 7.4 - Prob. 7.38QAPCh. 7.5 - Prob. 7.39QAPCh. 7.5 - Prob. 7.40QAPCh. 7.5 - In each of the following, identify the reactant...Ch. 7.5 - In each of the following, identify the reactant...Ch. 7.5 - Prob. 7.43QAPCh. 7.5 - Prob. 7.44QAPCh. 7.5 - Prob. 7.45QAPCh. 7.5 - Prob. 7.46QAPCh. 7.6 - Write all of the mole—mole factors for each of the...Ch. 7.6 - Write all of the mole—mole factors for each of the...Ch. 7.6 - The chemical reaction of hydrogen with oxygen...Ch. 7.6 - Prob. 7.50QAPCh. 7.6 - Carbon disulfide and carbon monoxide are produced...Ch. 7.6 - Prob. 7.52QAPCh. 7.7 - Sodium reacts with oxygen to produce sodium oxide....Ch. 7.7 - Nitrogen gas reacts with hydrogen gas to produce...Ch. 7.7 - Ammonia and oxygen react to form nitrogen and...Ch. 7.7 - Iron(III) oxide reacts with carbon to give iron...Ch. 7.7 - Nitrogen dioxide and water react to produce nitric...Ch. 7.7 - Calcium cyanamide, CaCN2 , reads with water to...Ch. 7.7 - Prob. 7.59QAPCh. 7.7 - When the gases dihydrogen sulfide and oxygen...Ch. 7.8 - Prob. 7.61QAPCh. 7.8 - Prob. 7.62QAPCh. 7.8 - Classify each of the following as exothermic or...Ch. 7.8 - Classify each of the following as exothermic or...Ch. 7.8 - Classify each of the following as exothermic or...Ch. 7.8 - Classify each of the following as exothermic or...Ch. 7.8 - a. What is meant by the rate of a reaction? b. Why...Ch. 7.8 - a. How does a catalyst affect the activation...Ch. 7.8 - Prob. 7.69QAPCh. 7.8 - How would each of the following change the rate of...Ch. 7 - Prob. 7.71UTCCh. 7 - Using the models of the molecules (black = C,...Ch. 7 - A dandruff shampoo contains dipyrithione,...Ch. 7 - Ibuprofen, an anti-inflammatory drug in Advil, has...Ch. 7 - Prob. 7.75UTCCh. 7 - Balance each of the following by adding...Ch. 7 - Prob. 7.77UTCCh. 7 - Prob. 7.78UTCCh. 7 - If blue spheres represent nitrogen atoms, purple...Ch. 7 - Prob. 7.80UTCCh. 7 - Prob. 7.81UTCCh. 7 - Prob. 7.82UTCCh. 7 - Calculate the molar mass for each of the...Ch. 7 - Calculate the molar mass for each of the...Ch. 7 - How many grams are in 0.150 mole of each of the...Ch. 7 - How many grams are in 2.25 moles of each of the...Ch. 7 - How many moles are in 25.0 g of each of the...Ch. 7 - How many moles are in 4.00 g of each of the...Ch. 7 - Identify the type of reaction for each of the...Ch. 7 - Identify the type of reaction for each of the...Ch. 7 - Prob. 7.91AQAPCh. 7 - Prob. 7.92AQAPCh. 7 - Prob. 7.93AQAPCh. 7 - Prob. 7.94AQAPCh. 7 - Identify each of the following as an oxidation or...Ch. 7 - Prob. 7.96AQAPCh. 7 - When ammonia NH3 gas reacts with fluorine gas, the...Ch. 7 - When nitrogen dioxide NO2 gas from car exhaust...Ch. 7 - Pentane gas, C5H12 undergoes combustion with...Ch. 7 - Prob. 7.100AQAPCh. 7 - Prob. 7.101AQAPCh. 7 - The equation for the formation of nitrogen oxide...Ch. 7 - Prob. 7.103CQCh. 7 - Prob. 7.104CQCh. 7 - Prob. 7.105CQCh. 7 - A toothpaste contains 0.240% by mass sodium...Ch. 7 - Prob. 7.107CQCh. 7 - Prob. 7.108CQCh. 7 - Prob. 7.109CQCh. 7 - Prob. 7.110CQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What type of reaction and how much energy is involved in a reaction of boron trichloride with hydrochloric acid? B2H6(g) +6Cl2(g) ⟷2BCl3(g) + 6HCl(g) ΔH= -755.4 kJ/mole Group of answer choices Exothermic with ΔH = -755.4 kJ/mole. Exothermic with ΔH = +755.4 kJ/mole. Endothermic with ΔH = -755.4 kJ/mole. Endothermic with ΔH = +0.001324 kJ/mole. Endothermic with ΔH = +755.4 kJ/mole.arrow_forwardA student reacted 1.01g salicylic acid with 2.06mL acetic anhydride and isolated 1.40g aspirin. A) Calculate moles of acetic anhydride used. B) Using the moles of limiting reactant present, calculate the theoretical yield of aspirin. C) Calculate the percent yield of the aspirin synthesis.arrow_forwardClayton akers has inhaled an estimate 6.00 * 10 to the 21th power molecules of 02 before delivering his fastball. How many moles of 02 is this?arrow_forward
- Initial Temp of HCl: 22.96 C Final Temp of HCl: 28.30 C Moles of HCl: .05 moles Moles of NaOH: .05 moles Cs water: 4.184 J/g C Accepted delta Hrxn: -55.8 kJ/mol 1. what is the qwater. pls include formula. 2. calculate delta Hrxnn using the moles, temp and cswater 3. calculate delta Hrxn % errorarrow_forwardIf the empirical formula of a compound is found to be CH2, which of the following value is a plausible molecular mass for the compound? The answer is 70.15. How did you get that?arrow_forwardGive answer to all parts? Initial Temp of HCl: 22.96 C Final Temp of HCl: 28.30 C Moles of HCl: .05 moles Moles of NaOH: .05 moles Cs water: 4.184 J/g C Accepted delta Hrxn: -55.8 kJ/mol 1. what is the qwater. pls include formula. 2. calculate delta Hrxnn using the moles, temp and cswater 3. calculate delta Hrxn % errorarrow_forward
- toluene can be oxidized by the oxygen gas in air, under controlled conditions to make benzoic acid. what is the percent yield of a reaction that converts 1.36 kg of toulene to 1.24 kg of benzoic acid? answer as a percent with 1 decimal place 2 C6H5CH3 + 3 O2 --> 2 C6H5CO2H + 2 H2Oarrow_forwardWhat mass of aspirin (C₉H₈O₄) is produced from 68.2 g of C₇H₆O₃ assuming 95.0% yield from the reaction below? C₇H₆O₃ (s) + C₄H₆O₃ (s) → C₉H₈O₄ (s) + HC₂H₃O₂ (aq).arrow_forward14. Romeo preserves a dozen of milkfish for his birthday celebration. He used 200g of NaCl to fully preserve it in just 3 days. How many moles of NaCl do the 200g have? A. 1. 42 mole NaCl B. 2. 42 mole NaCl C. 3. 42 mole NaCl D. 4. 42 mole NaClarrow_forward
- If Kc = 1.52 for O2 (g) + 2 CO (g) ⇌ 2 CO2 (g), what is the value of Kc for the reaction 4 CO2 (g) ⇌ 2 O2 (g) + 4 CO (g) (with 3 significant figures)? Choose one option. a. 1.32 b. 0.329 c. 3.04 d. 1.52 e. 0.433arrow_forwardHow many moles of MgCl2 are produced from 35.0g of Mg and 35.0g of HCl? Mg + 2HCl → MgCl2 + H2 Group of answer choices: 0.956 moles 1.44 moles 0.479 moles 1.92 molesarrow_forwardCalculate the value of Kc for the reaction: 2 NO(g) + Br2(g) ⇌ 2 NOBr (g) using the following information. Do NOT use scientific notation. 2 NO(g) ⇌ N2(g) + O2(g) Kc1 = 1 x 1030N2(g) + Br2(g) + O2(g) ⇌ 2 NOBr(g) Kc2 = 2 x 10-27arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY