
The ground-state electron configurations listed here are incorrect. Explain what mistakes have been made in each and write the correct electron configurations.
Al: 1s22s22p43s23p3
B: 1s22s22p5
F: 1s22s22p6
(a)
Interpretation:
The ground-state electron configurations of the given elements whose configurations made incorrect should be corrected.
Concept Introduction:
An orbital is an area of space in which electrons are orderly filled. The maximum capacity in any type of orbital is two electrons. An atomic orbital is defined as the region of space in which the probability of finding the electrons is highest. It is subdivided into four orbitals such as
There are three basic principles in which orbitals are filled by the electrons.
- 1. Aufbau principle: In German, the word 'aufbau' means 'building up'. The electrons are arranged in various orbitals in the order of increasing energies.
- 2. Pauli exclusion principle: An electron does not have all the four quantum numbers.
- 3. Hund’s rule: Each orbital is singly engaged with one electron having the maximum same spin capacity after that only pairing occurs.
The electron configuration is the allocation of electrons of an atom in atomic orbitals. Electronic configuration of a particular atom is written by following the three basic principles.
To find: Correct the ground-state electron configuration of
Answer to Problem 7.76QP
(a)
The correct ground-state electron configuration of 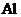 whose configuration made incorrect as
whose configuration made incorrect as
Explanation of Solution
All the 13 electrons are placed in the atomic orbitals by following Aufbau principle, Pauli exclusion principle and Hund’s rule.
All the 13 electrons of
There are two electrons present in each
(b)
Interpretation:
The ground-state electron configurations of the given elements whose configurations made incorrect should be corrected.
Concept Introduction:
An orbital is an area of space in which electrons are orderly filled. The maximum capacity in any type of orbital is two electrons. An atomic orbital is defined as the region of space in which the probability of finding the electrons is highest. It is subdivided into four orbitals such as
There are three basic principles in which orbitals are filled by the electrons.
- 1. Aufbau principle: In German, the word 'aufbau' means 'building up'. The electrons are arranged in various orbitals in the order of increasing energies.
- 2. Pauli exclusion principle: An electron does not have all the four quantum numbers.
- 3. Hund’s rule: Each orbital is singly engaged with one electron having the maximum same spin capacity after that only pairing occurs.
The electron configuration is the allocation of electrons of an atom in atomic orbitals. Electronic configuration of a particular atom is written by following the three basic principles.
To find: Correct the ground-state electron configuration of B whose configuration made incorrect as
Answer to Problem 7.76QP
(b)
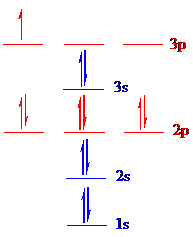
The correct ground-state electron configuration of
Explanation of Solution
All the 5 electrons are placed in the atomic orbitals by following Aufbau principle, Pauli exclusion principle and Hund’s rule.
All the 5 electrons of
There are two electrons present in each
(c)
Interpretation:
The ground-state electron configurations of the given elements whose configurations made incorrect should be corrected.
Concept Introduction:
An orbital is an area of space in which electrons are orderly filled. The maximum capacity in any type of orbital is two electrons. An atomic orbital is defined as the region of space in which the probability of finding the electrons is highest. It is subdivided into four orbitals such as
There are three basic principles in which orbitals are filled by the electrons.
- 1. Aufbau principle: In German, the word 'aufbau' means 'building up'. The electrons are arranged in various orbitals in the order of increasing energies.
- 2. Pauli exclusion principle: An electron does not have all the four quantum numbers.
- 3. Hund’s rule: Each orbital is singly engaged with one electron having the maximum same spin capacity after that only pairing occurs.
The electron configuration is the allocation of electrons of an atom in atomic orbitals. Electronic configuration of a particular atom is written by following the three basic principles.
To find: Correct the ground-state electron configuration of F whose configuration made incorrect as
Answer to Problem 7.76QP
(c)
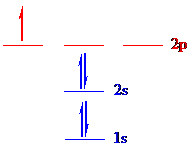
The correct ground-state electron configuration of
Explanation of Solution
All the 9 electrons are placed in the atomic orbitals by following Aufbau principle, Pauli exclusion principle and Hund’s rule.
All the 9 electrons of
There are two electrons present in each
Want to see more full solutions like this?
Chapter 7 Solutions
CHEMISTRY-ALEK 360 ACCES 1 SEMESTER ONL
Additional Science Textbook Solutions
General, Organic, and Biological Chemistry - 4th edition
Introductory Chemistry (5th Edition) (Standalone Book)
Thermodynamics, Statistical Thermodynamics, & Kinetics
Chemistry
General, Organic, & Biological Chemistry
Principles of General, Organic, Biological Chemistry
- In defining the sizes of orbitals, why must we use an arbitrary value, such as 90% of the probability of finding an electron in that region?arrow_forwardThe table below is partially completed for subshells A and B, where subshell B is the next subshell higher in energy compared to subshell A. For example, if subshell A is the 4s subshell, then subshell B would be the 3d subshell. Use this criteria and the information provided to complete the table.arrow_forwardWhat type of electron orbital (i.e., s, p, d, or f) is designated by an electron with quantum numbers (a) n=1,l=0,m l =0(b) n=3,l=2,m l =1? (c) n=4,l=3,m l =3arrow_forward
- As the weapons officer aboard the Srarship Chemistry, it is your duty to configure a photon torpedo to remove an electron from the outer hull of an enemy vessel. You know that the work function (the binding energy of the electron) of the hull of the enemy ship is 7.52 1019 J. a. What wavelength does your photon torpedo need to be to eject an electron? b. You find an extra photon torpedo with a wavelength of 259 nm and fire it at the enemy vessel. Does this photon torpedo do any damage to the ship (does it eject an electron)? c. If the hull of the enemy vessel is made of the element with an electron configura tion of [Ar]4s13d10, what metal is this?arrow_forward6.19 Fill in the blanks below to complete a description of the photoelectric effect experiment. (You should be able to do this with just one or two words in each blank.) A beam of_________ strikes __________ causing to be emitted.arrow_forwardSketch a picture of the 90% boundary surface of an s orbital and the px orbital. Be sure the latter drawing shows why the p orbital is labeled px and not py, for example.arrow_forward
- 6.96 When a helium atom absorbs light at 58.44 nm, an electron is promoted from the 1s orbital to a 2p orbital. Given that the ionization energy of (ground state) helium is 2372 kJ/ mol, find the longest wavelength of light that could eject an electron from the excited state helium atom.arrow_forward6.29 A mercury atom emits light at many wavelengths, two of which are at 435.8 and 546.1 nm. Both of these transitions are to the same final state. (a) What is the energy difference between the two states for each transition? (b) lf a transition between the two higher energy states could be observed, what would be the frequency of the light?arrow_forwardGive a brief wave description of light. What are two characteristics of light waves?arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





