
Concept explainers
As we will learn in Chapter 9, an epoxide is an ether with an oxygen atom in a three-membered ring.
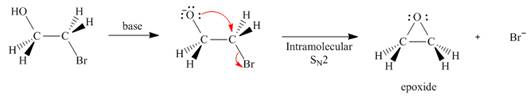
Assume that each of the following starting materials can be converted to an epoxide by this reaction. Draw the product formed (including stereochemistry) from each starting material. Why might some of these reactions be more difficult than others in yielding nucleophilic substitution products?
a. 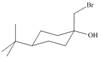 b.
b.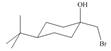 c.
c.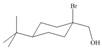 d.
d. 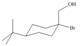
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
Connect Access Card For Organic Chemistry
Additional Science Textbook Solutions
Thermodynamics, Statistical Thermodynamics, & Kinetics
Chemistry: Matter and Change
Chemistry: The Central Science (14th Edition)
Inorganic Chemistry
Chemistry & Chemical Reactivity
Chemistry: The Molecular Nature of Matter
- Draw the products obtained in each of the following reactions, assuming that only one equivalent of each reagent is used in each case:arrow_forwardDraw the product of the following SN2 reaction, including the stereochemistry at all stereogenic centers. The product of this reaction is aprepitant, a drug used to treat nausea and emesis (vomiting) in chemotherapy patients.arrow_forwardGiven that an E2 reaction proceeds with anti periplanar stereochemistry, draw the products of each elimination. The alkyl halides in (a) and (b) are diastereomers of each other. How are the products of these two reactions related? Recall from Section 3.2A that C6H5 −is a phenyl group, a benzene ring bonded to another group.arrow_forward
- Given that an E2 reaction proceeds with anti periplanar stereochemistry, draw the products of each elimination. The alkyl halides in (a) and (b) are diastereomers of each other. How are the products of these two reactions related? Recall from Section 3.2A that C6H5– is a phenyl group, a benzene ring bonded to another group.arrow_forwardProvide the reagents necessary to accomplish each of the following transformation.arrow_forwardHere in Chapter 19, we learned that converting a ketone or aldehyde to an acetal is a good way to protect the carbonyl group, because an acetal is composed of ether linkages. In Problem 17.57, we saw that a ketone or aldehyde can be converted into an epoxide, which is a cyclic ether. Why would an epoxide be a poor choice as a protecting group?arrow_forward
- Draw the major organic product of the indicated reaction conditions. Do not show any by-products, just how the organic starting material is transformed.arrow_forwardThe following incomplete reaction sequences give a starting material and a product that can be prepared from it. Show the reagents taht would be necessary to carry out each transformation and any major products taht would be formed in the intermediate stages. There may be more than one correct way to complete a sequence. Thanks!arrow_forwardSynthesize the product shown from the starting materials, including intermediates formed at each steparrow_forward
- Identify reagents that can be used to achieve each of the following transformationsarrow_forwardWhat reagents should be used to carry out the following syntheses?arrow_forwardBe sure to answer all parts. Draw the product formed when the following alkene is treated with either [1] HBr alone and [2] HBr in the presence of peroxides. CH2CHCH2CH2CH2CH3arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





