
Macroscale and Microscale Organic Experiments
7th Edition
ISBN: 9781305577190
Author: Kenneth L. Williamson, Katherine M. Masters
Publisher: Brooks Cole
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 7, Problem 8Q
Interpretation Introduction
Interpretation:
A flow chart that depicts the separation procedure used for separation of mixture of toluic acid,
Concept introduction:
Separation of a mixture that contains an acidic, basic and neutral substance involves acid-base extraction technique.
The structure of toluic acid is as follows:
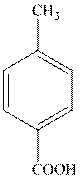
The structure of
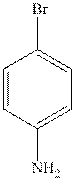
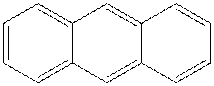
The structure of anthracene is as follows:
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Draw a flow sheet to show how you would separate the components of a mixture containing an acid substance, toluic acid, a basic substance, p-bromoaniline, and anthracene, a neutral substance.
Write an equation for the reaction of butylamine with HCI
What is the acid base reaction involved in the titration of liberated HCl (from the reaction of methylethyl ketone and hydroxylamine hydrochloride) and NaOH?
Chapter 7 Solutions
Macroscale and Microscale Organic Experiments
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 18-17 Hexanoic (caproic) acid has a solubility in water of about 1 g/100 mL water. Which part of the molecule contributes to water solubility, and which part prevents solubility?arrow_forwardWrite the chemical equation for the acid dissociation of acetaminophen, C8H9O2N. Write the Ka expression for the acid dissociation of acetaminophen.arrow_forwardWrite the equation for the hydrolysis of aniline (C6H5NH2), an organic base that smells like rotten fish.arrow_forward
- write the esterification reaction of lauric acid with ethanolarrow_forwardCreate a flowchart to demonstrate how to separate a butyric acid and hexane mixture.arrow_forward18-31 Formic acid is one of the components responsible for the sting of biting ants and is injected under the skin by bee and wasp stings. The pain can be relieved by rubbing the area of the sting with a paste of baking soda (NaHCO3) and water, which neutralizes the acid. Write an equation for this reaction.arrow_forward
- 17-35 Suppose that you take a bottle of benzaldehyde (a liquid, bp 179°C) from a shelf and find a white solid in the bottom of the bottle. The solid turns litmus red; that is, it is acidic. Yet aldehydes are neutral compounds. How can you explain these observations?arrow_forward18-29 Complete the equations for these acid—base reactions. (a) (b) (c) (d) (e)arrow_forward17-78 Complete the following equation for these reactions.arrow_forward
- 16-35 (Chemical Connections 16B ) What is an alkaloid? Are all alkaloids basic to litmus?arrow_forwardWrite an equation for the reaction of chloroacetic acid (Ka=1.5103) with trimethylamine (Kb=5.9105) . Calculate the equilibrium constant for the reaction. If 0.10 M solutions of these two species are mixed, what will be their concentrations at equilibrium?arrow_forward18-15 Draw a structural formula for the dimer formed when two molecules of formic acid interact by hydrogen bonding.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning