
Concept explainers
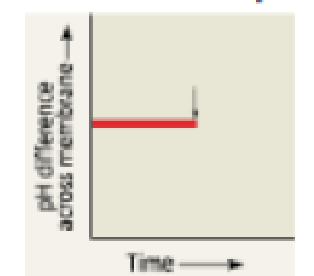
DRAW IT The graph here shows the pH difference across the inner mitochondrial membrane over time in an actively respiring cell. At the time indicated by the vertical arrow, a
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
Campbell Biology in Focus; Modified Mastering Biology with Pearson eText -- ValuePack Access Card -- for Campbell Biology in Focus (2nd Edition)
Additional Science Textbook Solutions
Laboratory Manual For Human Anatomy & Physiology
Biology: Life on Earth
Fundamentals of Anatomy & Physiology Plus Mastering A&P with eText - Access Card Package (10th Edition) (New A&P Titles by Ric Martini and Judi Nath)
Human Anatomy & Physiology
Microbiology with Diseases by Taxonomy (5th Edition)
Human Anatomy & Physiology (11th Edition)
- Figure 7.11 Dinitrophenol (DNP) is an "uncoupler" that makes the inner mitochondrial membrane "leaky" to protons. It was used until 1938 as a weight-loss drug. What effect would you expect DNP to have on the change in pH across the inner mitochondrial membrane? Why do you think this might be an effective weight-loss drug?arrow_forwardIn phase II of ETS diagram and label the ATP synthase along with the inner mitochondrial membrane it is sitting in. Your diagram must show the intermembrane space and the mitochondrial matrix. Describe how it works step by steparrow_forwardDraw and label a lipid bilayer containing the large mitochondrial trans-membrane protein complexes representing complex I, II, III, and IV, and ATP Synthase. Make two more of these drawings. Label the first one mitochondrial electron source and using a different ink color, indicate the source(s) of electrons. Label the second mitochondrial energy source, and indicate the source of energy driving the electron transport chain. In the third, label the mitochondrial electron acceptor in its proper position. In this third drawing, also include ATP synthase (in its correct orientation-the spherical part is on the opposite side of the membrane as the H+ reservoir). Label your diagrams with the correct names for the membranes, ATP synthase, and to show where the protons (H+) are pumped to drive ATP synthesis.arrow_forward
- Figure 7.11 Dinitrophenol (DNP) is an "uncoupler" that makes the inner mitochondrial membrane "leaky" to protons. It was used until 1938 as a weight- loss drug. What effect would you expect DNP to have on the change in pH across the inner mitochondrial membrane? Why do you think this might be an effective weight-loss drug? Intermembrane space Mitochondrial matrix ATP Synthase ADP Inner mitochondrial membrane ATP Figure 7.11 ATP synthase is a complex, molecular machine that uses a proton (H) gradient to form ATP from ADP and inorganic phosphate (Pi). (Credit: modification of work by Klaus Hoffmeier)arrow_forwardWhat is the major route for protons moving from the inter membrane space back into the mitochondrial matrix during oxidative phosphorylation? 1. Protons carried across the membrane by a molecule of dinitrophenol 2. Protons are transported along with ADP by the ATP/ADP translocase.3. Protons enter the half channel in subunit a facing the inter membrane space and exit via the half channel facing the mitochondrial matrix. Choose 1 correct answer explain? Give typing answer with explanation and conclusionarrow_forwardReferring to the figure below, explain why NADH yields more ATP than FADH2 does. Electron-transport and proton pump Oxidative phosphorylation Outer mitochondrial membrane H* -Intermembrane H+ H+ H+ space H* H+ H+ Cytochrome c H+ COQH, CoQ UU COQH2 CoQ JU U Inner mitochondrial membrane Ht e ATPase Complex II Complex II Complex IV Complex e ADP +P - Mitochondrial matrix NADH NAD+ FADH2 FAD АТР H+ -H+ H+ H20arrow_forward
- Look at the diagram of the mitochondrial electron transport chain below and answer the questions that follow Ubiquinone Membrane Z Complex I Complex III Y Complex I| Succinate Fumarate H,0 NAD NADH There are 2 names for the component in the orange circle labelled Y, they are: and The name of the component X in the red circle is The membrane labelled Z is the membrane Which component accepts electrons from complex 1?arrow_forwardAlthough the outer mitochondrial membrane is permeable to all small molecules, the inner mitochondrial membrane is essentially impermeable in the absence of specific transport proteins. Consider this information answer: The ATP generated by oxidative respiration is used throughout the cell. The majority of ATP production occurs in the mitochondrial matrix. How do you think ATP is made accessible to enzymes in the cytosol and other organelles?arrow_forwardFirst find and label ATP Synthase on the diagram below. Make boxes and add the labels for ATP, ADP, and Pi. Use an arrow indicating ATP being made by the right part of the synthase. This diagram also shows the mitochondrial electron transport chain. Work with your team to add the following labels Complexes l-IV Intermembrane space 02 FADH2 FAD H20 NAD+ Matrix NADH Path of electrons Protons Figure 7.10b Biology: How Life Works, Second Edition © 2016 Macmillan Educationarrow_forward
- You are studying the process of oxidative phosphorylation in the lab. You isolate several mitochondria from cells and place them in a buffered solution with a low pH and observe that the mitochondria begin to synthesize ATP. Which of the following is the best explanation for this observation? F It increases the conc. of OH-, causing the mitochondria to pump H+ to the intermembrane space. It increases the diffusion of H+ from the mitochondrial matrix to the intermembrane space. It increases the diffusion of H+ from the intermembrane space to the matrix. It increases the conc. of OH- in the mitochondrial matrix.arrow_forwardATP is synthesized from ADP, P, and a proton on the matrix side of the in- ner mitochondrial membrane. We will refer to the matrix side as the "inside" of the inner mitochondrial membrane (IMM). (a) H* transport from the outside of the IMM into the matrix drives this process. The pH inside the matrix is 8.2, and the outside is more acidic by 0.8 pH units. Assuming the IMM membrane potential is 168 mV (inside negative), calculate AG for the transport of 1 mol of H* across the IMM into the matrix at 37 °C: Houtside) Hinside) (b) Assume three mol H* must be translocated to synthesize one mol ATP by coupling of the following reactions: ADP + P, + Hinskde) ATP + H,O(ATP synthesis) 3Hinside)(proton transport) 3Houtside)arrow_forwardIn the 1930s, some physicians prescribed low doses of a compound called dinitrophenol (DNP) to help patients lose weight. This unsafe method was abandoned after some patients died. DNP uncouples the chemiosmotic machinery by making the lipid bilayer of the inner mitochondrial membrane leaky to H+. Chemical agents that cause this effect are called uncouplers. Explain how this could cause weight loss and also death. Considering the danger, is there any use for compounds like DNP or other uncouplers?arrow_forward
 Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning
Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning

