
ORGANIC CHEM ALC PKG
8th Edition
ISBN: 9780134595634
Author: Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Question
Chapter 7.6, Problem 13P
(a)
Interpretation Introduction
Interpretation:
The mechanism for the reaction of 2-butyne with one equivalent of bromine should be given.
Concept Introduction:
Addition of halides to
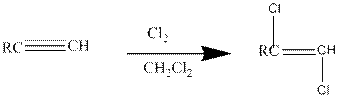
If excess halides are present, a second addition reaction also occurs.
(b)
Interpretation Introduction
Interpretation:
The configuration of the product obtained in the above reaction should be identified.
Concept Introduction:
Addition of halides to alkynes: Halogens like chlorine and bromine adds to the alkyne compounds. The general form of the reaction is given below.

If excess halides are present, a second addition reaction also occurs.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Drawing on what you know about the stereochemistry of alkene addition reactions, a. write the mechanism for the reaction of 2-butyne with one equivalent of Br2. b. predict the configuration of the product of the reaction.
1. Write down all the steps of the reaction of 2-Methyl-2-hexene with sulfuric acid and name the product formed.
Will provide helpful ratings for correct solution.
a. What is the major product obtained from the reaction of propene and Br2 plus excess Cl-? b. Indicate the relative amounts of the stereoisomers that are obtained.
Chapter 7 Solutions
ORGANIC CHEM ALC PKG
Ch. 7.1 - Prob. 1PCh. 7.1 - Name the following:Ch. 7.1 - What is the molecular formula for a monocyclic...Ch. 7.1 - Draw the condensed and skeletal structures for...Ch. 7.1 - Draw the structure and give the common and...Ch. 7.2 - Prob. 6PCh. 7.2 - Name the following:Ch. 7.3 - What orbitals are used to form the carbon-carbon ...Ch. 7.4 - Prob. 9PCh. 7.4 - Why does cis-2-butene have a higher boiling point...
Ch. 7.6 - Prob. 12PCh. 7.6 - Prob. 13PCh. 7.7 - Prob. 14PCh. 7.7 - Which alkyne should be used for the synthesis of...Ch. 7.7 - Prob. 16PCh. 7.8 - Prob. 17PCh. 7.8 - Only one alkyne forms an aldehyde when it...Ch. 7.9 - Describe the alkyne you should start with and the...Ch. 7.9 - Prob. 20PCh. 7.10 - Prob. 21PCh. 7.10 - Prob. 22PCh. 7.10 - Prob. 23PCh. 7.10 - Rank the following from strongest base to weakest...Ch. 7.10 - Prob. 26PCh. 7.12 - Prob. 28PCh. 7 - What is the major product obtained from the...Ch. 7 - Draw a condensed structure for each of the...Ch. 7 - A student was given the structural formula of...Ch. 7 - Prob. 32PCh. 7 - What is each compounds systematic name?Ch. 7 - What reagents should be used to carry out the...Ch. 7 - Prob. 35PCh. 7 - Draw the mechanism for the following reaction:Ch. 7 - Prob. 37PCh. 7 - Prob. 38PCh. 7 - What is the major product of the reaction of 1 mol...Ch. 7 - Answer Problem 39, parts a-b, using 2-butyne as...Ch. 7 - What is each compounds systematic name? a....Ch. 7 - What is the molecular formula of a hydrocarbon...Ch. 7 - a. Starting with 3-methyl 1-butyne, how can you...Ch. 7 - Prob. 44PCh. 7 - Which of the following pairs are keto-enol...Ch. 7 - Prob. 46PCh. 7 - Do the equilibria of the following acid-base...Ch. 7 - What is each compounds systematic name?Ch. 7 - What stereoisomers are obtained when 2-butyne...Ch. 7 - Prob. 50PCh. 7 - Draw the keto tautomer for each of the following:Ch. 7 - Show how each of the following compounds can be...Ch. 7 - A chemist is planning to synthesize 3-octyne by...Ch. 7 - Prob. 54PCh. 7 - What stereoisomers are obtained from the following...Ch. 7 - Prob. 56PCh. 7 - Prob. 57PCh. 7 - Prob. 58PCh. 7 - Show how the following compound can be prepared...Ch. 7 - Prob. 60P
Knowledge Booster
Similar questions
- Is the reaction of 2-butene with HBr regioselective?arrow_forward2. In the bromination reactions, what is the function of CCl4? Why can it fulfil its role?3. Bromination proceeds by either free radical substitution or electrophillic addition. Based on Table 3, which mechanism is followed by alkanes? by alkenes? by alkynes?4. For which hydrocarbon type is light necessary for bromination to take place?arrow_forwardWhat are the colors of bromine and potassium permanganate? Write the chemical equations involved in the reaction of cyclohexene with the following: a.) Br2 b.) cold KMnO4arrow_forward
- Phineas and Ferbs, two brothers who enjoy vacations, doing fun things every summer. This summer the brothers and their friends carry out an organic synthesis with an unknown compound (L1) that contains 52% Carbon, 6% Hydrogen and 42% bromine, this compound (L1) is treated with magnesium in ether to obtain L2 , which reacts violently with D2O for 1-methyl cyclohexene with a deuterium atom in the methyl group (L3). The L2 reaction is treated with acetone followed by hydrolysis to give L4. Heating L4 with concentrated sulfuric acid gives L5, which decolors the bromine, obtaining L6. L5 undergoes hydrogenation with excess hydrogen and platinum as a catalyst giving rise to isobutyl cyclohexane. Determine the structures of compounds L1 through L6.arrow_forwardDraw the carbocation you'd expect from the reaction of 2-chloro-3-methylbutane with AlCl3?arrow_forwardReaction of HBr with 2-methylpropene yields 2-bromopropane. What is the structure of the carbocation intermediate formed during the reaction? Show the mechanism of the reaction.arrow_forward
- Why does bromine's color disappear when Br2 is added to an alkene sample?arrow_forwardReaction of hbr with 2-methylpropene yields 2 bromo 2 methylpropane. What is the structure of the carbocation formed during the reaction? Show the mechanism of reaction.arrow_forward1) What is the major product for the reaction of 3-bromo1-butene and HBr? Give the name of the product. 2) For this product, how many chiral carbon(s) are there in the structure?3) How many stereoisomers could we write for this product? 4) Write the Fischer Projections of all stereoisomers. (attach a file or other ways you could show your work)arrow_forward
- 2. In the bromination reactions, what is the function of CCl4? Why can it fulfil its role?3. Bromination proceeds by either free radical substitution or electrophillic addition. Based on Table 3, which mechanism is followed by alkanes? by alkenes? by alkynes?4. For which hydrocarbon type is light necessary for bromination to take place?5. What is the function of light in the bromination reaction? Why are alkenes and alkynes not included as samples?arrow_forwardOf the four possible cis,trans isomers, one is formed in over 85% yield. Q.) Which is the structure of the isomer formed in 85% yield? How do you account for its formation? Create a model to help you make this prediction.arrow_forwardChlorination of 2-butanone yields two isomeric products, each having the molecular formula C4H7ClO. (a) What are these two compounds? (b) Write structural formulas for the enol intermediates that lead to each of these compounds. (c) Using curved arrows, show the flow of electrons in the reaction of each of the enols with Cl2.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
