
Concept explainers
Interpretation:
The pH values after the addition of each proportion of the base to the acid is to be determined. The titration curve needs to be drawn.
Concept introduction:
A strong acid or a strong base can completely dissociate into its ions when they are in an aqueous solution. Sodium hydroxide is a strong base while nitric acid is a strong acid. When these two chemical species are reacted with each other, they form their ionic forms and a neutral solution.
Explanation of Solution
Initial pH of the analyte solution;
Nitric acid is a strong acid that completely dissociates in water.
The amount of hydrogen ion will be equal to the concentration of nitric acid thus,
The pH of the solution will be:
Addition of
Total amount of acid to be neutralized
Amount of base added
Therefore, the amount of acid remaining in the solution
The concentration of hydrogen ions
The pH of the solution
Addition of
Total amount of acid to be neutralized
Amount of base added
Therefore, the amount of acid remaining in the solution
The concentration of hydrogen ions
The pH of the solution
Addition of
Total amount of acid to be neutralized
Amount of base added
Therefore, the amount of acid remaining in the solution
The concentration of hydrogen ions
The pH of the solution
Addition of
Total amount of acid to be neutralized
Amount of base added
Therefore, the amount of acid remaining in the solution
The concentration of hydrogen ions
The pH of the solution
Addition of
Total amount of acid to be neutralized
Amount of base added
Therefore, the amount of acid remaining in the solution
The concentration of hydrogen ions
The pH of the solution
Addition of
Total amount of acid to be neutralized
Amount of base added
Therefore, the amount of acid remaining in the solution
The concentration of hydrogen ions
The pH of the solution
Addition of
Total amount of acid to be neutralized
Amount of base added
Therefore, the amount of acid remaining in the solution
The concentration of hydrogen ions
The pH of the solution
Addition of
Total amount of acid to be neutralized
Amount of base added
Therefore, the amount of acid remaining in the solution will be:
Since, both acid and base completely react to form salt and water, the pH of the solution
Addition of
Total amount of acid to be neutralized
Amount of base added
Therefore, the amount of excess base in the solution
The concentration of hydroxide ions
Thenthe pH of the solution can be calculated as follows:
Addition of
Total amount of acid to be neutralized
Amount of base added
Therefore, the amount of excess base in the solution
The concentration of hydroxide ions
Then the pH of the solution can be calculated as follows:
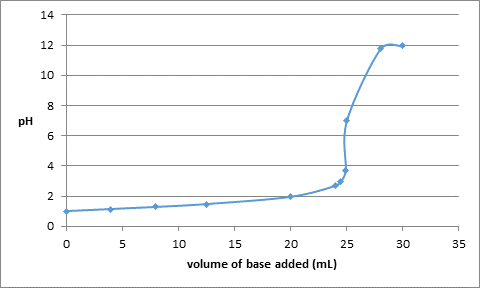
The data obtained from the calculations is as follows:
| Volume of base added (mL) | pH |
| 0.0 | 1 |
| 4.0 | 1.14 |
| 8.0 | 1.28 |
| 12.5 | 1.47 |
| 20.0 | 1.95 |
| 24.0 | 2.69 |
| 24.5 | 2.99 |
| 24.9 | 3.69 |
| 25.0 | 7.0 |
| 28.0 | 11.75 |
| 30.0 | 11.95 |
The titration curve can be drawn as follows:
Want to see more full solutions like this?
Chapter 8 Solutions
CHEM.PRINC.W/OWL2+REBATE+2 SUPPL.>IP<
- Repeat the procedure in Exercise 61, but for the titration of 25.0 mL of 0.100 M pyridine with 0.100 M hydrochloric acid (Kb for pyridine is 1.7 109). Do not calculate the points at 24.9 and 25.1 mL.arrow_forwardHow many grams of HI should be added to 265 mL of 0.215 M HCI so that the resulting solution has a pH of 0.38? Assume that the addition of HI does not change the volume of the resulting solution.arrow_forwardCalculate the pH during the titration of 50.00 mL of 0.100 M Sr(OH)2 with 0.100 M HNO3 after 0, 50.00, 100.00, and 150.00 mL nitric acid have been added. Graph the titration curve and compare with the titration curve obtained in Exercise 16.22.arrow_forward
- A certain acetic acid solution has pH = 2.68. Calculate the volume of 0.0975 M KOH required to reach the equivalence point in the titration of 25.0 mL of the acetic acid solution.arrow_forwardRepeat the procedure in Exercise 61, but for the titration of 25.0 mL of 0.100 M propanoic acid (HC3H5O2,Ka = 1.3 105) with 0.100 M NaOH.arrow_forwardCalculate the volume of 1.50 102 M NaOH that must be added to 500.0 mL of 0.200 M HCl to give a solution that has pH = 2.15.arrow_forward
- A 0.400-g sample of propionic acid was dissolved in water to give 50.0 mL of solution. This solution was titrated with 0.150 M NaOH. What was the pH of the solution when the equivalence point was reached?arrow_forwardCalculate the pH after 0.020 mole of HCl is added to 1.00 L of each of the four solutions in Exercise 22.arrow_forwardWhich of the solutions in Exercise 21 shows the least change in pH upon the addition of acid or base? Explain.arrow_forward
- A 0.400-M solution of ammonia was titrated with hydrochloric acid to the equivalence point, where the total volume was 1.50 times the original volume. At what pH does the equivalence point occur?arrow_forwardA solution is made by diluting 25.0 mL of concentrated HCl (37% by weight; density = 1.19 g/mL) to exactly 500 mL. Calculate the pH of the resulting solution.arrow_forwardWhat volume of 0.120 M NaOH must be added to 100. mL of 0.100 M NaHC2O4 to reach a pH of 4.70?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning





