
Organic Chemistry, 12e Study Guide/Student Solutions Manual
12th Edition
ISBN: 9781119077329
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8, Problem 15PP
Practice Problem 8.15
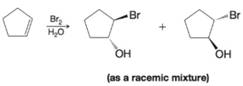
Write a mechanism to explain the following reaction.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
O PRACTICE PROBLEM 8.14
Starting with any needed alkene (or cycloalkene) and assuming you have deuterioace-
tic acid (CH3CO,D) available, outline syntheses of the following deuterium-labeled
compounds.s el en
olad lo nohibbs ad
CH3
(a) (CH3)2CHCH2CH,D
(b) (CH3),CHCHDCH3 (c)
(+ enantiomer)
(d) Assuming you also have available BD3:THF and CH3CO2T, can you suggest a
synthesis of the following?
hab erl
(+ enantiomer)he
imo
(nwond-ben)
CH3
H.
(asoholea)
Problems
8.29 Give the mechanistic symbols (SN1, SN2, E1, E2) that are most consistent with each of the
following statements:
(a) Methyl halides react with sodium ethoxide in ethanol only by this mechanism.
(b) Unhindered primary halides react with sodium ethoxide in ethanol mainly by this
mechanism.
(c) When cyclohexyl bromide is treated with sodium ethoxide in ethanol, the major prod-
uct is formed by this mechanism.
(d) The substitution product obtained by solvolysis of tert-butyl bromide in ethanol arises
by this mechanism.
(e) In ethanol that contains sodium ethoxide, tert-butyl bromide reacts mainly by this
mechanism.
(f) These reaction mechanisms represent concerted processes.
(g) Reactions proceeding by these mechanisms are stereospecific.
(h) These reaction mechanisms involve carbocation intermediates.
(i) These reaction mechanisms are the ones most likely to have been involved when the
products are found to have a different carbon skeleton from the substrate.
(j) Alkyl…
8.30 Outline an efficient synthesis of each of the following compounds from the indicated start-
ing material and any necessary organic or inorganic reagents:
(a) Cyclopentyl cyanide from cyclopentane
(b) Cyclopentyl cyanide from cyclopentene
(c) Cyclopentyl cyanide from cyclopentanol
(d) NCCH₂CH₂CN from ethyl alcohol
(e) Isobutyl iodide from isobutyl chloride
(f) Isobutyl iodide from tert-butyl chloride
(g) Isopropyl azide from isopropyl alcohol
(h) Isopropyl azide from 1-propanol
(i) (S)-sec-Butyl azide from (R)-sec-butyl alcohol
(i) (S)-CH3CH₂CHCH3 from (R)-sec-butyl alcohol
SH
Chapter 8 Solutions
Organic Chemistry, 12e Study Guide/Student Solutions Manual
Ch. 8 - Prob. 1PPCh. 8 - Prob. 2PPCh. 8 - Practice Problem 8.3 Provide mechanistic...Ch. 8 - Prob. 4PPCh. 8 - Prob. 5PPCh. 8 - Prob. 6PPCh. 8 - Prob. 7PPCh. 8 - Prob. 8PPCh. 8 - Prob. 9PPCh. 8 - Prob. 10PP
Ch. 8 - Prob. 11PPCh. 8 - Prob. 12PPCh. 8 - Practice Problem 8.13
Specify the appropriate...Ch. 8 - Prob. 14PPCh. 8 - Practice Problem 8.15 Write a mechanism to explain...Ch. 8 - Prob. 16PPCh. 8 - Prob. 17PPCh. 8 - Prob. 18PPCh. 8 - Practice Problem 8.19 Treating cyclohexene with l,...Ch. 8 - Prob. 20PPCh. 8 - Practice Problem 8.21
Predict the products of the...Ch. 8 - Prob. 22PPCh. 8 - Prob. 23PPCh. 8 - Prob. 24PPCh. 8 - Prob. 25PPCh. 8 - Write structural formulas for the products that...Ch. 8 - Prob. 27PCh. 8 - Prob. 28PCh. 8 - 8.29. Give the structure of the products that you...Ch. 8 - Give the structure of the products you would...Ch. 8 - Prob. 31PCh. 8 - Prob. 32PCh. 8 - Prob. 33PCh. 8 - Prob. 34PCh. 8 - Prob. 35PCh. 8 - Prob. 36PCh. 8 - Prob. 37PCh. 8 - When 3, 3-dimethyl-2-butanol is neared with...Ch. 8 - Prob. 39PCh. 8 - Prob. 40PCh. 8 - Prob. 41PCh. 8 - Prob. 42PCh. 8 - 8.43. Write a mechanism that explains the...Ch. 8 - 8.44. Write a mechanism for the following...Ch. 8 - Write a mechanism that explains formation of the...Ch. 8 - Prob. 46PCh. 8 - 8.47. Farnesene (below) is a compound found in the...Ch. 8 - Limonene is a compound found in orange oil and...Ch. 8 - Prob. 49PCh. 8 - Synthesize the following compound starting with...Ch. 8 - Predict features of their IR spectra that you...Ch. 8 - Deduce the structures of compounds A, B, and C,...Ch. 8 - Ricinoleic acid, a compound that can be isolated...Ch. 8 - 8.54. There are two dicarboxylic acids with the...Ch. 8 - Prob. 55PCh. 8 - Prob. 56PCh. 8 - Prob. 57PCh. 8 - Prob. 58PCh. 8 - 8.59. For each of the following questions, please...Ch. 8 - Prob. 60PCh. 8 - Prob. 61PCh. 8 - Prob. 62PCh. 8 - Triethylamine, (C2H5)3N, like all amines, has a...Ch. 8 - (a) Synthesize (3S, 4R)-3,...Ch. 8 - Prob. 2LGPCh. 8 - Prob. 3LGPCh. 8 - Prob. 4LGP
Additional Science Textbook Solutions
Find more solutions based on key concepts
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
1. What did each of the following scientists contribute to our knowledge of the atom?
a. William Crookes
b. E...
Chemistry For Changing Times (14th Edition)
Taking as references pure liquid sulfuric acid at 77°F and pure liquid water at 32°F and without using Figure 8...
Elementary Principles of Chemical Processes, Binder Ready Version
Write Lewis symbols for the following atoms. (a) Kr; (b) Ge; (c) N; (d) Ga; (e) As; (f) Rb.
General Chemistry: Principles and Modern Applications (11th Edition)
Determine whether each molecule is polar or nonpolar. ClO3- SCl2 SCl4 BrCl5
Chemistry: A Molecular Approach
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- • PRACTICE PROBLEM 8.13 Specify the appropriate alkene and reagents for synthesis of each of the following alcohols by hydroboration–oxidation. (a) (c) OH (e) CH3 OH AH OH no mobe OH ( (b) (d) (f) OH HT H3 D OH OH HO OHarrow_forwardProvide the reagents and solvents (where appropriate) needed to bring about the following transformations. (a) CI (b)arrow_forward12.6 (opq) Predict the major product and give the name of the reactionarrow_forward
- 8.30 Outline an efficient synthesis of each of the following compounds from the indicated starting material and any necessary organic or inorganic reagents: (g) Isopropyl azide from isopropyl alcohol (h) Isopropyl azide from 1-propanol (i) (S)-sec-butyl azide from (R)-sec-butyl alcoholarrow_forward4.33 Select the compound in each of the following pairs that will be converted to the corresponding alkyl bromide more rapidly on being treated with hydrogen bromide. Explain the reason for your choice. (a) 1-Butanol or 2-Butanol (b) 2-Methyl-1-butanol or 2-butanol (c) 2-Methyl-2-butanol or 2-butanol (d) 2-Methylbutane or 2-butanol (e) 1-Methylcyclopentanol or cyclohexanol Draw the energy diagrams of an SN1 reaction and an SN2 reaction. Include in your drawing anexample reaction. Identify the rate limiting step and label it as unimolecular or bimolecular.arrow_forward4.2 Propose a mechanism for the following reaction and account for the formation of a racemic mixture. H3O+ HOarrow_forward
- • PRACTICE PROBLEM 8.12 Treating a hindered alkene such as 2-methyl-2-butene with BH3:THE leads to the for- mation of a dialkylborane instead of a trialkylborane. When 2 mol of 2-methyl-2-butene is added to 1 mol of BH3, the product formed is bis(3-methyl-2-butyl)borane, nicknamed in reagent "disiamylborane." Write its structure. Bis(3-methyl-2-butyl)borane is a useful certain syntheses that require a sterically hindered borane. (The name “disiamyl" comes from "disecondary-iso-amyl," a completely unsystematic and unacceptable name. The name "amyl" is an old common name for a five-carbon alkyl group.) donbyarrow_forward• PRACTICE PROBLEM 8.24 A, B, and C are alkynes. Elucidate their structures and that of D using the following reaction roadmap. H2, Pt H,, Pt A (C3H14) (C3H14) IR: 3300 cm (1) O3 (2) HOẠC HO, H2, Pt (C3H12) (C3H16) (1) O3 (2) HOAC hol bian vd beeollot Но. ОН AOHarrow_forward8.23 In each of the following indicate which reaction will occur faster. Explain your reasoning. (D) Solvolysis of 1-bromo-2,2-dimethylpropane or tert-butyl bromide in ethanol (E) Solvolysis of isobutyl bromide or sec-butyl bromide in aqueous formic acid (F) Reaction of 1-chlorobutane with sodium acetate in acetic acid or with sodium methoxide in methanolarrow_forward
- 5.34 Select the compound in each of the following pairs that will be converted to the corresponding alkyl bromide more rapidly on being treated with hydrogen bromide. Explain the reason for your choice. (a) 1-Butanol or 2-butanol (b) 2-Methyl-1-butanol or 2-butanol (c) 2-Methyl-2-butanol or 2-butanol (d) 2-Methylbutane or 2-butanol dovolonentanol or cyclohexanolarrow_forwardPredict the reagents for reactions A and B.arrow_forwarddon't know Which of the following, when reacted with HCl, would result in the formation of the same major product at both low and high temperatures? (a) (b)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY