
Concept explainers
Write Lewis dot symbols for the following atoms and ions:
Interpretation:
The Lewis dot symbols for the given atoms and ions are to be written.
Concept introduction:
Lewis dot symbols contain dots that give information about the valence electrons.
Lewis dot symbols show both bond and lone pairs as dots.
Lewis structures show bonds as lines and lone pairs as dots.
Answer to Problem 5QP
Solution:
a)
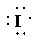
b)
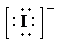
c)
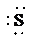
d)
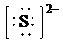
e)
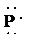
f)
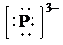
g)
h)
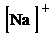
i)
j)
k)
l)
m)
n)
Explanation of Solution
a) Atom—
The number of valence electrons in
atom is
So, there are
So, the Lewis structure for
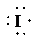
b) Ion—
The number of valence electrons in
So, there are
So, the Lewis structure for
is as follows:
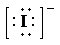
c) Atom—
The number of valence electrons in
atom is
So, there are 3 lone pairs present.
So, the Lewis structure for
is as follows:
d) Ion—
The number of valence electrons in
So, there are
So, the Lewis structure for
is as follows:
e) Atom—
The number of valence electrons in
atom is
So, there are
So, the Lewis structure for
is as follows:
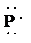
f) Ion—
The number of valence electrons in
So, there are
So, the Lewis structure for
is as follows:
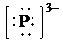
g) Atom—
The number of valence electrons in
atom is
So, there are no lone pairs present.
So, the Lewis structure for
is as follows:
h) Ion—
The number of valence electrons in
So, the Lewis structure for
is as follows:
i) Atom—
The number of valence electrons in
atom is
So, the Lewis structure for
is as follows:
j) Ion—
The number of valence electrons in
So, the Lewis structure for
is as follows:
k) Atom—
The number of valence electrons in
atom is
So, the Lewis structure for
is as follows:
l) Ion—
The number of valence electrons in
So, the Lewis structure for
is as follows:
m) Atom—
The number of valence electrons in
atom is
So, the Lewis structure for
is as follows:
n) Ion—
The number of valence electrons in
So, the Lewis structure for
is as follows:
Want to see more full solutions like this?
Chapter 8 Solutions
Package: Chemistry With Aleks 360 Access Card, 2-semester
- Using the bond dissociation enthalpies in Table 8.8, estimate the enthalpy of combustion of gaseous methane, CH4, to give water vapor and carbon dioxide gas.arrow_forwardDefine the term lattice energy. Why, energetically, do ionic compounds form? Fig. 3-8 illustrates the energy changes involved in the formation of MgO(s) and NaF(s). Why is the lattice energy of MgO(s) so different from that of NaF(s)? The magnesium oxide is composed of Mg2+ and O2 ions. Energetically, why does Mg2+O2 form and not Mg+O? Why doesnt Mg3+O3 form?arrow_forwardWrite a Lewis symbol for atom or ion. Mg2+arrow_forward
- Which of the following atoms would be expected to form negative ions in binary ionic compounds and which would be expected to form positive ions: P, I, Mg, Cl, In, Cs, O, Pb, Co?arrow_forwardWrite the Lewis symbols for Ca2+ and Cl-arrow_forwardDraw the Lewis structure for the nitrate ion, NO3-, with formal charges on each atom and resonance structures.arrow_forward
- Write Lewis dot symbols for the following atoms and ions. (a) I (b) I− (c) S (d) S2−arrow_forwardWrite Lewis structures for the following: (c) C2F6 (contains a C¬C bond), (d) AsO3 3 -, (e) H2SO3 (H is bonded to O), (f) NH2Cl.. Arrange the bonds in each of the following sets in order of increasing polarity: (a) C¬F, O¬F, Be¬F; (b) O¬Cl, S¬Br, C¬P; (c) C¬S, B¬F, N¬O. What is the Lewis symbol for each of the following atoms or ions? (a) K, (b) As, (c) Sn2 + , (d) N3 Write electron configurations for the following ions and determine which have noble-gas configurations: (a) Cd2+, (b) P3-, (c) Zr4+arrow_forwardIn which of the compounds CH4, NH3, H2O, SiH4, or H2S is δ+ for hydrogen the greatest? In which one does hydrogen bear a partial negative charge?arrow_forward
- The magnitude of the lattice energy (energy required to separate the ions) for the compounds K2O and CaO are shown below with respect to Coulombic attraction, which is the best explanation for the difference in lattice energy?arrow_forwardWhich of the following ionic compounds would be expected to have the highest lattice energy? RbBr, KF, NaI, MgBr2arrow_forwardDraw Lewis structures for these ions and show which atom in each bears the formal charge. Q.) HCO3-arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning




