
Concept explainers
Interpretation:
- The products of the given reaction have to be predicted.
Concept Introduction:
Resonance Contributor: The appropriate structure with the localized electrons is called a resonance contributor, a resonance structure, or a contributing resonance structure.
Delocalized electrons: The sharing of electrons between two or more atoms known as delocalization of electrons. In order to have delocalized electrons, the system must be planar and have alternative double bonds and single bonds.
Resonance hybrid: The actual structure with delocalized electrons is called a resonance hybrid.
Answer to Problem 61P
Correct Answer:
The compounds A, B, C, E, F, L, M, N, and O have delocalized electrons.
Explanation of Solution
Reason for correct options:
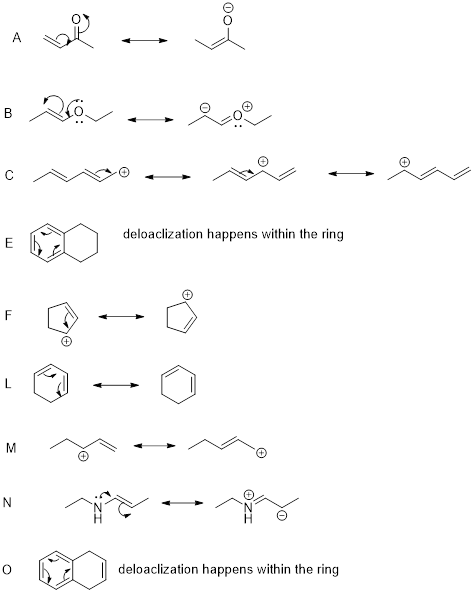
The above compounds all have delocalized electrons, which undergo electron movement. The resonance structures can be drawn as shown above.
Hence, the correct options are A, B, C, E, F, L, M, N, and O.
Reason for in-correct options:
The remaining compounds do not contain delocalized electrons, conjugated system is absent.
Hence, the in-correct options are D, G, H, I, J, and K.
- The compounds containing delocalized electrons were predicted.
Want to see more full solutions like this?
Chapter 8 Solutions
Study Guide And Solutions Manual For Organic Chemistry, Global Edition
- The number of allylic positions in compound A is ?arrow_forwardWhy alkyl groups considered as an ortho- , para-directing?arrow_forwardDraw the resonance structure of the ff. and identify if aromatic or nonaromatic through Huckel’s rule. note: I need the answer immediately. I will send a good rate right away as well.arrow_forward
- a) Predict the major product of the reaction and identify the type of reaction (substitution or addition). Explain the function of Pt used in the reaction. (with First image) b) Write the reaction of the mononitration of chlorobenzene and predict the major products with reasons. c) Identify all the carbon atoms in the following compounds that are sp2 (Circle the carbon(s) your selected on image two)arrow_forwardWhich carbocation would be lowest energy? Options: A B C Darrow_forwardDraw each hybridisation steps (with detailed explanation) of this reaction pleasearrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
