
Concept explainers
(a)
Interpretation:
A suitable reagent should be predicted for the given transformations.
Concept introduction:
Elimination reaction: In this reaction, two substituents are removed from the substrate to give the product in presence of base.
Anti-Markovnikov’s rule: unsymmetrical
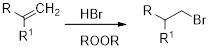
(b)
Interpretation:
A suitable reagent should be predicted for the given transformations.
Concept introduction:
Hydrobromination:
A hydrobromination reaction is one of the electrophilic additions to alkenes to yield the corresponding bromo

(c)
Interpretation:
A suitable reagent should be predicted for the given transformations.
Concept introduction:
Osmium tetra oxide:
Cis-alkene undergoes oxidation using osmium tetra oxide followed by hydrogen peroxide hydrolysis which furnish cis 1,2
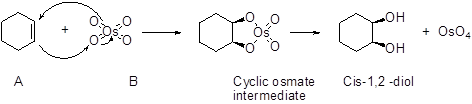
(d)
Interpretation:
A suitable reagent should be predicted for the given transformations.
Concept introduction:
Elimination reaction: In this reaction, two substituents are removed from the substrate to give the product in presence of base.
Anti-dihydroxylation: It is a process of adding two –OH groups from different planes to the alkene. Alkenes are treated with peroxy acids followed by aqueous acids and gives anti-dihydroxylation products.
Syn-dihydroxylation: It is a process of adding two –OH groups from the same plane to the alkene. Potassium per manganite and osmium tetroxide etc, are used as reagents.
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
ORGANIC CHEMISTRY-PRINT COMPANION (LL)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





