
CHEMISTRY: ATOMS FIRST VOL 1 W/CONNECT
14th Edition
ISBN: 9781259327933
Author: Burdge
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8, Problem 8.10QP
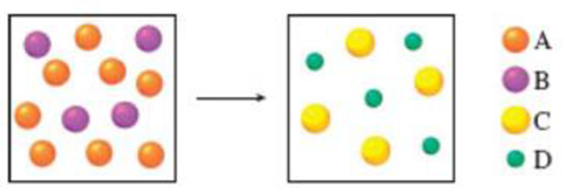
Which of the following equations best represents the reaction shown in the diagram?
(a) 8A + 4B → C + D
(b) 4A + 8B → 4C + 4D
(c) 2A + B → C + D
(d) 4A + 2B → 4C + 4D
(e) 2A + 4B → C + D
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
CHEMISTRY: ATOMS FIRST VOL 1 W/CONNECT
Ch. 8.1 - Write and balance the chemical equation for the...Ch. 8.1 - Write and balance the chemical equation that...Ch. 8.1 - Prob. 1PPBCh. 8.1 - Prob. 1PPCCh. 8.1 - Butyric acid (also known as butanoic acid,...Ch. 8.1 - Another compound found in milk fat that appears to...Ch. 8.1 - Prob. 2PPBCh. 8.1 - Prob. 2PPCCh. 8.1 - Prob. 8.3WECh. 8.1 - Prob. 3PPA
Ch. 8.1 - Using the chemical species A2, B, and AB, write a...Ch. 8.1 - Prob. 3PPCCh. 8.1 - Prob. 8.1.1SRCh. 8.1 - Prob. 8.1.2SRCh. 8.1 - Prob. 8.1.3SRCh. 8.1 - Prob. 8.1.4SRCh. 8.1 - Prob. 8.1.5SRCh. 8.2 - Combustion of a 5.50-g sample of benzene produces...Ch. 8.2 - The combustion of a 28.1-g sample of ascorbic acid...Ch. 8.2 - Prob. 4PPBCh. 8.2 - Prob. 4PPCCh. 8.2 - Prob. 8.2.1SRCh. 8.2 - Prob. 8.2.2SRCh. 8.2 - Prob. 8.2.3SRCh. 8.3 - Prob. 8.5WECh. 8.3 - Nitrogen and hydrogen react to form ammonia...Ch. 8.3 - Prob. 5PPBCh. 8.3 - Prob. 5PPCCh. 8.3 - Prob. 8.6WECh. 8.3 - Calculate the mass of water produced by the...Ch. 8.3 - Prob. 6PPBCh. 8.3 - The models here represent the reaction of nitrogen...Ch. 8.3 - Prob. 8.3.1SRCh. 8.3 - Prob. 8.3.2SRCh. 8.3 - Prob. 8.3.3SRCh. 8.3 - Prob. 8.3.4SRCh. 8.4 - Alka-Seltzer tablets contain aspirin, sodium...Ch. 8.4 - Ammonia is produced by the reaction of nitrogen...Ch. 8.4 - Prob. 7PPBCh. 8.4 - The diagrams show a reaction mixture before and...Ch. 8.4 - Aspirin, acetylsalicylic acid (C9H8O4), is the...Ch. 8.4 - Diethyl ether is produced from ethanol according...Ch. 8.4 - What mass of ether will be produced if 207 g of...Ch. 8.4 - The diagrams show a mixture of reactants and the...Ch. 8.4 - Prob. 8.4.1SRCh. 8.4 - Prob. 8.4.2SRCh. 8.4 - Prob. 8.4.3SRCh. 8.4 - Prob. 8.4.4SRCh. 8.4 - Prob. 8.4.5SRCh. 8 - Prob. 8.1QPCh. 8 - Prob. 8.2QPCh. 8 - Why must a chemical equation he balanced? What law...Ch. 8 - Write an unbalanced equation to represent each of...Ch. 8 - Prob. 8.5QPCh. 8 - Prob. 8.6QPCh. 8 - For each of the following unbalanced chemical...Ch. 8 - Prob. 8.8QPCh. 8 - Balance the following equations using the method...Ch. 8 - Which of the following equations best represents...Ch. 8 - Prob. 8.11QPCh. 8 - Determine whether each of the following equations...Ch. 8 - Prob. 8.13QPCh. 8 - Prob. 8.14QPCh. 8 - Prob. 8.15QPCh. 8 - Prob. 8.16QPCh. 8 - Prob. 8.17QPCh. 8 - Prob. 8.18QPCh. 8 - Prob. 8.19QPCh. 8 - Prob. 8.20QPCh. 8 - Prob. 8.21QPCh. 8 - Prob. 8.22QPCh. 8 - Prob. 8.23QPCh. 8 - On what law is stoichiometry based? Why is it...Ch. 8 - Prob. 8.25QPCh. 8 - Prob. 8.26QPCh. 8 - Prob. 8.27QPCh. 8 - Prob. 8.28QPCh. 8 - Prob. 8.29QPCh. 8 - Prob. 8.30QPCh. 8 - Prob. 8.31QPCh. 8 - Prob. 8.32QPCh. 8 - Prob. 8.33QPCh. 8 - When copper(II) sulfate pentahydrate (CuSO4 5H2O)...Ch. 8 - For many years, the extraction of gold from other...Ch. 8 - Prob. 8.36QPCh. 8 - Nitrous oxide (N2O) is also called laughing gas....Ch. 8 - Prob. 8.38QPCh. 8 - Prob. 8.39QPCh. 8 - Prob. 8.1VCCh. 8 - Prob. 8.2VCCh. 8 - Prob. 8.3VCCh. 8 - Prob. 8.4VCCh. 8 - Prob. 8.40QPCh. 8 - Prob. 8.41QPCh. 8 - Why is the theoretical yield of a reaction...Ch. 8 - Why is the actual yield of a reaction almost...Ch. 8 - Prob. 8.44QPCh. 8 - Prob. 8.45QPCh. 8 - Reactants A (red) and B (blue) combine in the...Ch. 8 - Prob. 8.47QPCh. 8 - Prob. 8.48QPCh. 8 - Prob. 8.49QPCh. 8 - Propane (C3H8) is a minor component of natural gas...Ch. 8 - Prob. 8.51QPCh. 8 - Prob. 8.52QPCh. 8 - Prob. 8.53QPCh. 8 - Prob. 8.54QPCh. 8 - Prob. 8.55QPCh. 8 - Prob. 8.56QPCh. 8 - Disulfur dichloride (S2Cl2) is used in the...Ch. 8 - Prob. 8.58QPCh. 8 - Prob. 8.59QPCh. 8 - Prob. 8.60QPCh. 8 - Prob. 8.61QPCh. 8 - Prob. 8.62QPCh. 8 - Prob. 8.63QPCh. 8 - Prob. 8.64QPCh. 8 - Prob. 8.65QPCh. 8 - Industrially, nitric acid is produced by the...Ch. 8 - Prob. 8.67QPCh. 8 - Prob. 8.68QPCh. 8 - Prob. 8.69QPCh. 8 - Prob. 8.70QPCh. 8 - Prob. 8.71QPCh. 8 - Prob. 8.72QPCh. 8 - Prob. 8.73QPCh. 8 - Prob. 8.74QPCh. 8 - Prob. 8.75QPCh. 8 - Prob. 8.76QPCh. 8 - Prob. 8.77QPCh. 8 - Prob. 8.78QPCh. 8 - Prob. 8.79QPCh. 8 - The combustion of a 5.50-g sample of oxalic acid...Ch. 8 - Prob. 8.81QPCh. 8 - Prob. 8.82QPCh. 8 - Prob. 8.83QPCh. 8 - Prob. 8.84QPCh. 8 - Prob. 8.85QPCh. 8 - Prob. 8.86QPCh. 8 - Potash is any potassium mineral that is used for...Ch. 8 - A 21.496-g sample of magnesium is burned in air to...Ch. 8 - Prob. 8.89QPCh. 8 - Prob. 8.90QPCh. 8 - Prob. 8.91QPCh. 8 - Prob. 8.92QPCh. 8 - Prob. 8.93QPCh. 8 - Prob. 8.94QPCh. 8 - Prob. 8.95QPCh. 8 - Prob. 8.96QPCh. 8 - Prob. 8.97QPCh. 8 - Prob. 8.98QPCh. 8 - A compound X contains 63.3 percent manganese (Mn)...Ch. 8 - Calculate the mass of water produced in the...Ch. 8 - Calcium phosphide (Ca3P2) and water react to form...Ch. 8 - Prob. 8.3KSPCh. 8 - Prob. 8.4KSP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The pictures below show a molecular-scale view of a chemical reaction between the compounds AB2 and B2. (Green balls represent B atoms and orange balls are A atoms). The box on the left represents the reactants at the instant of mixing, and the box on the right shows what is left once the reaction has gone to completion. Was there a limiting reactant in this reaction? If so, what was it? Write a balanced chemical equation for this reaction. As usual, your equation should use the smallest possible whole number coefficients for all substances.arrow_forwardA 100.0-g mixture made up of NaCl03, Na2CO3, NaCl, and NaHCO3 is heated, producing 5.95 g of oxygen, 1.67 g of water, and 14.5 g of carbon dioxide. NaCl does not react under the conditions of the experiment. The equations for the reactions that take place are: 2NaClO3(s)2NaCl(s)+3O2(g)Na2CO3(s)2Na2O(s)+CO2(g)2NaHCO3(s)Na2O(s)+2CO2(g)+H2O Assuming 100% decomposition of NaClO3, Na2CO3, and NaHCO3, what is the composition of the mixture in grams?arrow_forward4.68 The pictures below show a molecular-scale view of a chemical reaction between the compounds AB2 and B2. (Green balls represent B atoms and orange balls are A atoms). The box on the left represents the reactants at the instant of mixing, and the box on the right shows what is left once the reaction has gone to completion. Was there a limiting reactant in this reaction? If so, what was it? Write a balanced chemical equation for this reaction. As usual, your equation should use the smallest possible whole number coefficients for all substances.arrow_forward
- 3.75 The following pictures show a molecular-scale view of a chemical reaction between the compounds AB2 and B2. (A atoms are shown in blue and B atoms in white). The box on the left represents the reactants at the instant of mixing, and the box on the right shows what is left once the reac- tion has gone to completion. Write a balanced chemical equation for this reaction. As usual, your equation should use the smallest possible whole number coefficients for all substances.arrow_forward4.106 An ore sample with a mass of 670 kg contains 27.7% magnesium carbonate, MgCO3. If all of the magnesium carbonate in this ore sample is decomposed to form carbon dioxide, describe how to determine what mass of CO2 is evolved during the process.arrow_forwardThe pictures below show a molecular-scale view of a chemical reaction between H2 and CO to produce methanol, CH3OH The box on the left represents the reactants at the instant of mixing, and the box on the right shows what is left once the reaction has gone to completion. D Was there a limiting reactant in this reaction? If so, what was it? Write a balanced chemical equation for this reaction. As usual, your equation should use the smallest possible whole number coefficients for all substances.arrow_forward
- Many cereals are made with high moisture content so that the cereal can be formed into various shapes before it is dried. A cereal product containing 58% H2O by mass is produced at the rate of 1000. kg/h. What mass of water must be evaporated per hour if the final product contains only 20.% water?arrow_forwardFig. 5-5 illustrates a schematic diagram of a combustion device used to analyze organic compounds. Given that a certain amount of a compound containing carbon, hydrogen, and oxygen is combusted in this device, explain how the data relating to the mass of CO2 produced and the mass of H2O produced can be manipulated to determine the empirical formula.arrow_forwardUsing solid circles for H atoms and open circles for O atoms, make a drawing that shows the molecular level representation for the balanced equation of H2 and O2 reacting to form H2O.arrow_forward
- Many cereals are made with high moisture content so that the cereal can be formed into various shapes before it is dried. A cereal product containing 58% H2O by mass is produced at the rate of 1000. kg/h. What mass of water must be evaporated per hour if the final product contains only 20.% water?arrow_forwardWrite an equation from the following description: reactants are gaseous NH3 and O2, products are gaseous NO2 and liquid H2O, and the stoichiometric coefficients are 4, 7, 4, and 6, respectively.arrow_forward3.18 Diborane and related compounds were proposed as rocket fuels in the 1950s. A representative reaction for this class of molecules is that of B2H6 and O2 to form B2O3 and H2O. Write the balanced chemical equation for this process.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY