
Concept explainers
(a)
Interpretation:
The formulas of the simplest compounds formed by the reaction of hydrogen with silicon, germanium, tin, and lead has to be determined.
Concept Introduction:
Valence electrons are outer shell electrons that take part in the
Chemical properties of an element can be determined by the number of valence electrons.
The elements with same valence electrons have similar chemical and physical properties and are placed in the same group in periodic table.
The periodic table is given below:
Group 4A in the periodic table contains 4 valence electrons and the outer electronic configuration of halogens is
Hydrides are binary compounds formed by hydrogen with another element.
(b)
Interpretation:
Concept Introduction:
According to physical and chemical properties, the elements can be further divided into metals, non-metals and metalloids.
In a group, the metallic character of an element increases from top to bottom whereas in a period, it decreases from left to right.
Ionic compounds are formed by the transfer of electrons between a metal and a nonmetal. Molecular (covalent) compounds are formed by the sharing of electrons between two nonmetals.
(c)
Interpretation:
The reaction between radium
Concept Introduction:
Valence electrons are outer shell electrons that take part in the chemical bond formation of an atom.
Chemical properties of an element can be determined by the number of valence electrons.
The elements with same valence electrons have similar chemical and physical properties and are placed in the same group in periodic table.
The periodic table is given below:
Group 2A in the periodic table contains 2 valence electrons and the outer electronic configuration of halogens is
Alkaline earth metals react with water to produce a metal hydroxide and molecular hydrogen.
(d)
Interpretation:
Metal in Group 2A would you expect to exhibit similar properties has to be given and the reason for this has to be explained.
Concept Introduction:
Diagonal relationship is the trend observed in the chemical behavior of a certain pairs of elements that are diagonally adjacent in different periods and groups in the periodic table.
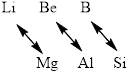
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
Chemistry
- Until the early 1960s the group 8A elements were called the inert gases; before that they were called the rare gases. The term rare gases was dropped after it was discovered that argon accounts for roughly 1% of Earth’s atmosphere. (a) Why was the term inert gases dropped? (b) What discovery triggered this change in name? (c)What name is applied to the group now?arrow_forwardThe elements sodium, aluminum, and chlorine are in the same period.(a) Which has the greatest electronegativity?(b) Which of the atoms is smallest?(c) Write the Lewis structure for the simplest covalent compound that can form between aluminum and chlorine.(d) Will the oxide of each element be acidic, basic, or amphoteric?arrow_forwardThe first three ionization energies of an element X are 900, 1745, and 14912 kJ·mol–1. What is the most likely formula for the stable ion of X?arrow_forward
- (a) Which poisonous gas is evolved when white phosphorus is heated with Cone. NaOH solution? Write the chemical equation. (b) Write the formula of first noble gas compound prepared by N. Bartlett. What inspired N. Bartlett to prepare this compound? (c) Fluorine is a stronger oxidising agent than chlorine. Why? (d)Write one use of chlorine gas.arrow_forwardThe oxygen and nitrogen families have some obvious similarities and differences.(a) State two general physical similarities between Group 5A(15) and 6A(16) elements.(b) State two general chemical similarities between Group 5A(15) and 6A(16) elements.(c) State two chemical similarities between P and S.(d) State two physical similarities between N and O.(e) State two chemical differences between N and O.arrow_forwardComplete and balance the following equations. Write the entire equation, including the reactants and products, and include the physical state of each species. (a) Na(s) + O2(g)→ (b) K(s) + O2(g)→arrow_forward
- Chlorine reacts with oxygen to form Cl2O7. (a) What is thename of this product (see Table 2.6)? (b) Write a balancedequation for the formation of Cl2O71l2 from the elements.(c) Would you expect Cl2O7 to be more reactive towardH+1aq2 or OH-1aq2? (d) If the oxygen in Cl2O7 is consideredto have the -2 oxidation state, what is the oxidation state ofthe Cl? What is the electron configuration of Cl in this oxidationstate?arrow_forwardElement X is a metal with a valency 2. Element Y is a non-metal with a valency 3 (a) Write equations to show how X and Y form ions. (b) If Y is a diatomic gas, write the equation for the direct combination of X and Y to form a compound.arrow_forwardA certain element, M, is a main-group metal that reacts with chlorine to give a compound with the chemical formula MCl2 and with oxygen to give the compound MO.(a) To which group in the periodic table does element M belong?(b) The chloride contains 44.7% chlorine by mass. Name the element Marrow_forward
- (a) List four physical characteristics of a solid metal. (b) List two chemical characteristics of a metallic element.arrow_forward(a) One of the alkali metals reacts with oxygen to form a solidwhite substance. When this substance is dissolved in water,the solution gives a positive test for hydrogen peroxide, H2O2.When the solution is tested in a burner flame, a lilac-purpleflame is produced. What is the likely identity of the metal?(b) Write a balanced chemical equation for the reaction ofthe white substance with water.arrow_forwardThe oxygen and nitrogen families have some obvious sim-ilarities and differences.(a) State two general physical similarities between Group5A(15) and 6A(16) elements. (b) State two general chemical similarities between Group5A(15) and 6A(16) elements.(c) State two chemical similarities between P and S.(d) State two physical similarities between N and O.(e) State two chemical differences between N and O.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning


