
On the same graph, plot the effective nuclear charge (see Section 8.3) and atomic radius (see Figure 8.5) versus
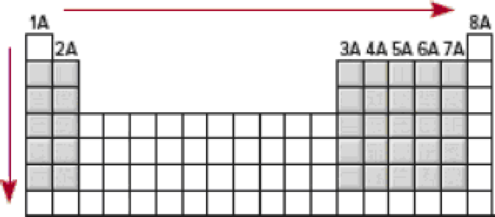
The increase in effective nuclear charge from left to right across a period and from top to bottom in a group for representative elements.
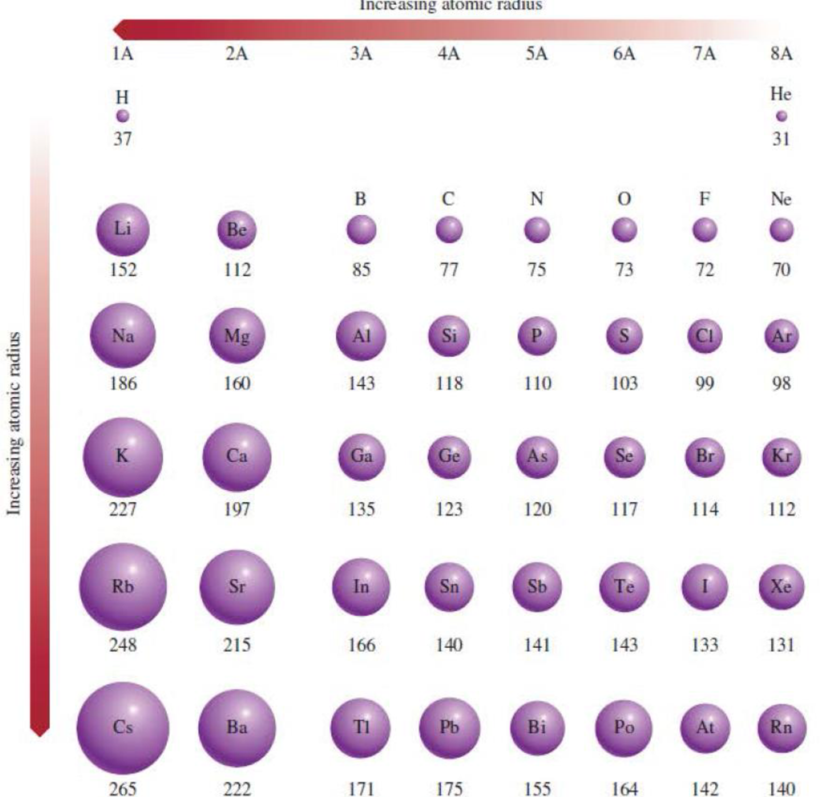
Figure 8.5 Atomic radii (in picometers) of representative elements according to their
positions in the periodic table. Note that there is no general agreement on the size of atomic radii. We focus only on the trends in atomic radii, not on their precise values.
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
Chemistry
Additional Science Textbook Solutions
Principles of Chemistry: A Molecular Approach (3rd Edition)
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Chemistry: The Molecular Nature of Matter
Living By Chemistry: First Edition Textbook
Organic Chemistry (8th Edition)
- Consider the figure in Chapter 7 of your textbook which shows the periodic trends in electron affinity. When you look across a period (row), why are the electron affinities of the Group 4A elements more negative than the electron affinities of the Group 5A elements? Group of answer choices The ratio of bonding radius to first ionization energy is smaller in 4A Atomic radii of the Group 4A elements are bigger than their corresponding group 5A elements Group 4A elements are more electronegative than the elements of Group 5A Group 5A half-filled p-subshells discourage the addition of an electron None of these choices explain the questionarrow_forwardRank these ions according to ionic radius. Rb+, As3-, Se2-, Br-, Sr2+ From largest radius to smallest radiusarrow_forwardRank the following four elements in order of decreasing atomic radius. Rank from largest to smallest radius. To rank items as equivalent, overlap them. Mg, Be, Sr, Caarrow_forward
- True or False. If false, give a reason. The 2nd ionization energy of magnesium is greater than that of sodium? the 1st ionization energy for a Group 13 element is actually less than for the adjacent Group 2. The electronic configuration of Cl is 1s2, 2s2, 2p6, 3s1, 3p6. Noble gases have the lowest ionization energies in their respective periods. The radii of the following elements are arranged as follows F < Li < C < Be < N < O < B Carbon has no d-orbital and thus pπ - dπ bonding is possible Solid peroxides formed by Group 1 elements only H2O2 acts as a strong oxidizing agent and a strong acid Negative ions are bigger than the corresponding atom.arrow_forwardWhich statement is true about effective nuclear charge?a) Effective nuclear charge decreases as you move to the right across a row in the periodic table.b) Effective nuclear charge increases as you move to the right across a row in the periodic table.c) Effective nuclear charge remains constant as you move to the right across a row in the periodic table.d) Effective nuclear charge increases and decreases at regular intervals as you move to the right across a row in the periodic table.arrow_forwardPlease solve all questions, including the subparts. What trend in first ionization energy occurs as you go up a group on the periodic table? Explain why this occurs. What trend in atomic radius occurs as you go up a group on the periodic table? Explain why this occurs. Arrange the following in order of increasing electronegativity: Br, F, I, Cl Write four quantum numbers to describe the highest energy electron in the magnesium atom. Be sure to include the four symbols and four correct numbers. Arrange the following in order of increasing first ionization energy: Br, F, I, Clarrow_forward
- Which of these statements is false? Second ionization energies are smaller than first ionization energies. Ionization energies increase on going from left to right across a period. Atomic radius increases on going down a group. A negative ion is larger than its parent neutral atom. Valence electrons are easier to remove than core electrons.arrow_forward3. The last orbital of element Y’s electronic configuration is defined by the following set of quantum numbers: n = 3, l = 1. It has 3 valence electrons.a. What is the element?b. What is its magnetic quantum number? (Clue: remember it has 3 valence electrons) c. Draw the Lewis structure of its chloride compounds and predict their shapes based on VSEPRtheory.arrow_forwardDescribe the trends of the element in a periodic table below and explain your answer: i)Electron affinity of group VII ii)Atomic radius of period 2 iii) Explain the trend for the first ionization energy based on the Table 1 below.arrow_forward
- Arrange the atoms in order of increasing atomic size. W, Ag, Fr, Te Arrange the atoms in order of increasing metallic property. S, F, Au, Sr List the following elements in order of: A. decreasing electron affinity - V, Ba, O, He, Al B. increasing ionization energy - Sr, Be, Ca, Mg C. increasing electronegativity - Ni, Pd, Ge, Rbarrow_forwardArrange the following in order of increasing ionic radius: I, Cs+, and Te2+. Explain this order. (You may use a periodic table.)arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning





