
Concept explainers
Interpretation: By using the formation of water from hydrogen and oxygen, the terms chemical reaction, reactant and product are to be explained.
Concept Introduction:
Chemical reaction is a process in which one substance breaks into two or more substances react with each other in the presence of reagent or catalyst under the appropriate experimental conditions leading to the formation of one or more substances. During
The substances undergoing a reaction is known as reactants, whereas the substances obtained from chemical reaction are called as products.
To Explain: The terms chemical reaction, reactant and product using the formation of water from hydrogen and oxygen.
Explanation of Solution
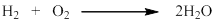
Formation of water takes place by the combination of hydrogen and oxygen.
- Chemical reaction is a combination of two or more substance to give one or more substance as product. Here, in the formation of water, hydrogen and oxygen combines with each other to give water.
- Hydrogen and oxygen undergoing the reaction are called as reactants.
- Water obtained from the reaction of hydrogen and oxygen is called as product.
By using the formation of water from hydrogen and oxygen, the terms chemical reaction, reactant and product are explained.
Want to see more full solutions like this?
Chapter 8 Solutions
Chemistry: Atoms First (Looseleaf)-Package
- Atomic masses are relative masses. What does this mean?arrow_forwardDetermine a chemical formula from elemental analysis (i.e., from % composition).arrow_forwardopical hydrocortisone is often used to treat a variety of skin conditions, such as insect bites, eczema, and rashes. Each molecule of hydrocortisone contains 21 atoms of carbon (plus other atoms). The mass percentage of carbon in hydrocortisone is 69.5 8%. What is the molar mass of hydrocortisone?arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning  Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER





