
Concept explainers
Interpretation:
Concept introduction:
Halogenation of
Radical chain reaction:
Initiation reaction: 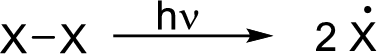
Chain propagation: 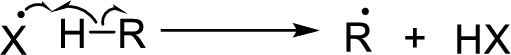
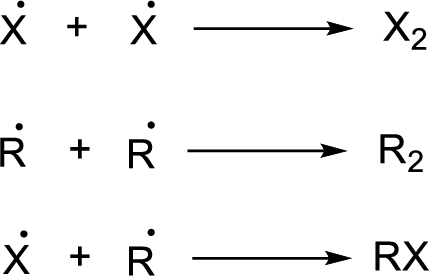
Chain termination:
It is a change in enthalpy of a homolysis reaction at absolute zero where a molecule is broken down into two free radicals.
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
OWL V2 with MindTap Reader and Student Solutions Manual eBook for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
- Give the process of Determining the Mechanism and Stereochemistry inNucleophilic Substitution ?arrow_forwardA) Provide the reagent and reaction mechanism to show how the reactants and products in the following reaction can interconvert B) under what conditions would the reaction I) favour reactants, II) favour the products and III) why?arrow_forwardIdentify the pericyclic reactions in the followingreaction schemes. Give the complete reaction name and indicate the course of the reaction with the aid of the arrow notation.arrow_forward
- 2) The Diels-Alder reaction, developed by German chemists Otto Diels and Kurt Alder (who received the Nobel Prize in 1950 for their discovery), has great synthetic importance due to the possibility of forming an unsaturated six-membered cycle without involving intermediates ionic. About the reaction, answer: (a) Indicate the reagents necessary for the synthesis of the following compounds, indicating who is the diene and who is the dienophile.arrow_forwardClassify the following sigmatropic rearrangement and determine whether it takes place readily under thermal or photochemical reaction conditions.arrow_forwardFuran undergoes electrophilic aromatic substitution more readily than benzene; mild reagents and conditions are sufficient.For example, furan reacts with bromine to give 2-bromofuran.(a) Propose mechanisms for the bromination of furan at the 2-position and at the 3-position. Draw the resonance forms ofeach sigma complex, and compare their stabilities.(b) Explain why furan undergoes bromination (and other electrophilic aromatic substitutions) primarily at the 2-position.O Br O123furan 2-bromofuranarrow_forward
- Identify the pericyclic reactions in the followingreaction schemes. Give the complete reactionname and indicate the course of the reaction withthe aid of the arrow notation.arrow_forwardStyrene (vinylbenzene) undergoes electrophilic aromatic substitution much faster thanbenzene, and the products are found to be primarily ortho- and para-substituted styrenes.Use resonance forms of the intermediates to explain these results.arrow_forwardGive the structure of compounds A to F in the following series of reactionsarrow_forward
- Which of the following series of reactions is a viable route form the starting material to the product shown below? The correct route is option B but please inlcude a detailed description explaining why and a step by step process:)arrow_forwardPredict the products of the following reduction reactions, including stereochemistry where needed. If the reaction product is racemic, indicate that by writing “racemic”. please explain stepsarrow_forwardGive the order of reactivity of carboxylic acid and its derivatives. What is thesignificance of this order of reactivity in predicting their interconversions?arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
