
(a)
Interpretation: All constitutional isomers formed in the given
Concept introduction: The removal of halide and neighboring
Answer to Problem 8.33P
All constitutional isomers formed in the given
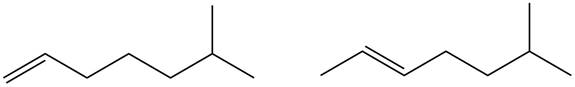
Figure 1
Disubstituted alkene is the major product of the reaction.
Explanation of Solution
The
In the given compound, two
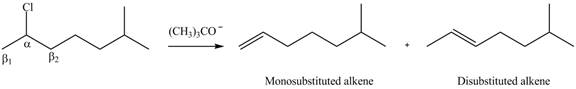
Figure 2
According to Zaitsev rule, more substituted alkene is obtained as a major product in
All constitutional isomers formed in the given
(b)
Interpretation: All constitutional isomers formed in the given
Concept introduction: The removal of halide and neighboring
Answer to Problem 8.33P
All constitutional isomers formed in the given
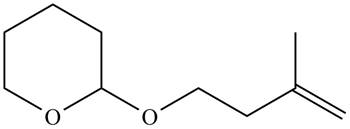
Figure 3
Monosubstituted alkene is the major product of the reaction.
Explanation of Solution
The
In the given compound, only one
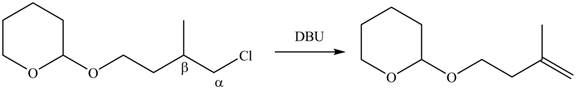
Figure 4
According to Zaitsev rule, more substituted alkene is obtained as a major product in
All constitutional isomers formed in the given
(c)
Interpretation: All constitutional isomers formed in the given
Concept introduction: The removal of halide and neighboring
Answer to Problem 8.33P
All constitutional isomers formed in the given
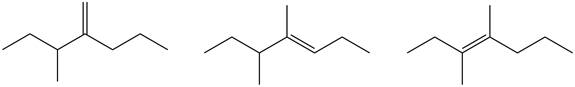
Figure 5
Tetrasubstituted alkene is the major product of the reaction.
Explanation of Solution
The
In the given compound, three
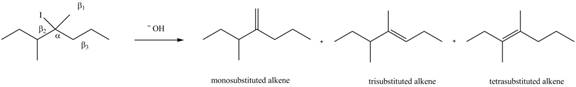
Figure 6
According to Zaitsev rule, more substituted alkene is obtained as a major product in
All constitutional isomers formed in the given
(d)
Interpretation: All constitutional isomers formed in the given
Concept introduction: The removal of halide and neighboring
Answer to Problem 8.33P
All constitutional isomers formed in the given
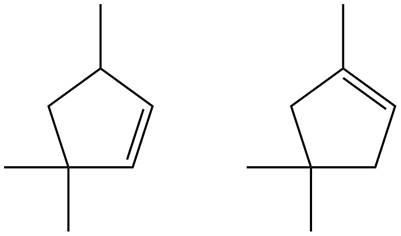
Figure 7
Trisubstituted alkene is the major product of the reaction.
Explanation of Solution
The
In the given compound, two
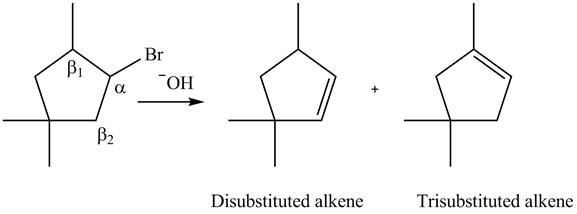
Figure 8
According to Zaitsev rule, more substituted alkene is obtained as a major product in
All constitutional isomers formed in the given
Want to see more full solutions like this?
Chapter 8 Solutions
ORGANIC CHEMISTRY SOLUTIONS MANUAL
- Sulfur ylides, like the phosphorus ylides, are usefulintermediates in organic synthesis. Methyl trans-chrysanthemate, anintermediate in the synthesis of the insecticide pyrethrin I,can be prepared from diene A and a sulfur ylide. Draw a stepwisemechanism for this reaction.arrow_forward(a) Are compounds B–D identical to or an isomer of A? (b) Give the IUPAC name for A.arrow_forwardConsider carbonyl compounds A–E drawn below. (a) Rank A–E in order of increasing stability. (b) Rank A–E in order of increasing amount of hydrate formed when treated with aqueous acid. (c) Which compound is most reactive in nucleophilic addition?arrow_forward
- Draw a stepwise mechanism for the following reaction, which forms the four-membered ring in azelnidipine, a drug used as a calcium channel blocker sold in Japan.arrow_forwardOne step in the synthesis of the antihistamine fexofenadine involves acid-catalyzed hydration of the triple bond in A. Draw a stepwise mechanism for this reaction and explain why only ketone B is formed.arrow_forwardProvide the missing starting material, reactant or product. Show appropriate stereochemistry. a) b)arrow_forward
- Draw a stepwise mechanism for the following Robinson annulation. This reaction was a key step in a synthesis of the steroid cortisone by R. B. Woodward and co-workers at Harvard University in 1951.arrow_forwardselect the expected major organic product.arrow_forwardDraw a stepwise mechanism for the attached reaction, which involves two Friedel–Crafts reactions. B was an intermediate in the synthesis of the antidepressant sertralinearrow_forward
- Draw a stepwise mechanism for the following reaction, a key step in the synthesis of the anti-inammatory drug celecoxib (trade name Celebrex).arrow_forwardDevise a stepwise synthesis of attached compound from dicyclopentadieneusing a Diels–Alder reaction as one step. You may also use organiccompounds having ≤ 4 C's, and any required organic or inorganicreagents.arrow_forward(−)-Hyoscyamine, an optically active drug used to treat gastrointestinal disorders, is isolated from Atropa belladonna, the deadly nightshade plant, by a basic aqueous extraction procedure. If too much base is used during isolation, optically inactive material is isolated. (a) Explain this result by drawing a stepwise mechanism. (b) Explain why littorine, an isomer isolated from the tailflower plant in Australia, can be obtained optically pure regardless of the amount of base used during isolation.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





