
Concept explainers
(a)
Interpretation:
The detailed mechanisms for the given reaction occurring via
Concept introduction:
The
In case of
Answer to Problem 8.44P
The
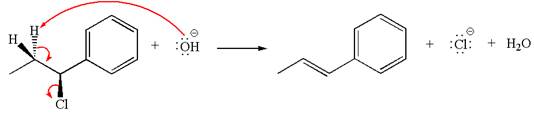
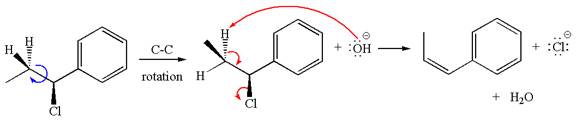
The
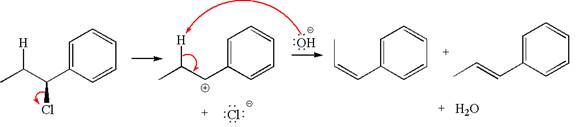
Explanation of Solution
The given reaction equation is:
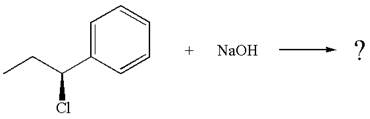
In the given reaction,
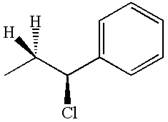
The hydrogen atom, indicated with the dash bond, is anti to
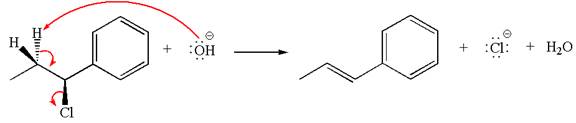
If the hydrogen atom, indicated with wedge bond, tends to eliminate, it must orient anti to
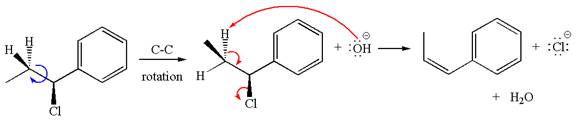
In
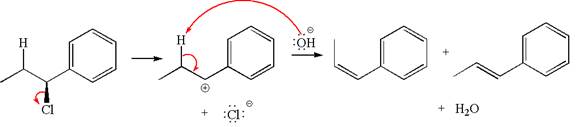
The products formed for the given reaction from both
(b)
Interpretation:
The detailed mechanisms for the given reaction occurring via
Concept introduction:
The
In case of
Answer to Problem 8.44P
The
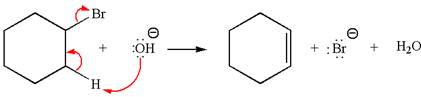
The
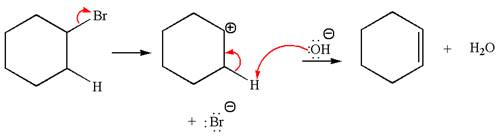
Explanation of Solution
The given reaction equation is:
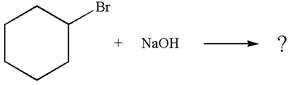
In the given reaction,
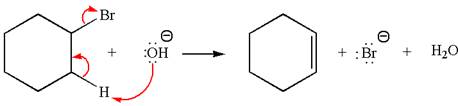
In
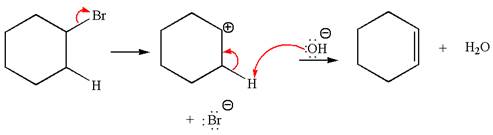
The products formed for the given reaction from both
(c)
Interpretation:
The detailed mechanisms for the given reaction occurring via
Concept introduction:
The
In case of
Answer to Problem 8.44P
The
The
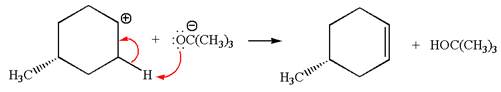
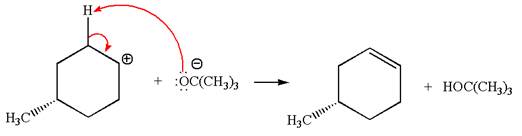
Explanation of Solution
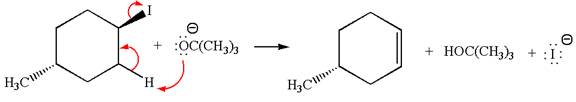
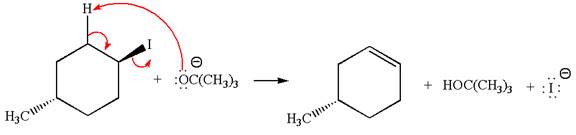
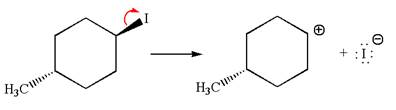
The given reaction equation is:
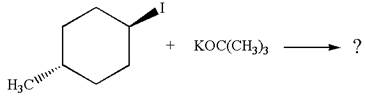
In the given reaction,
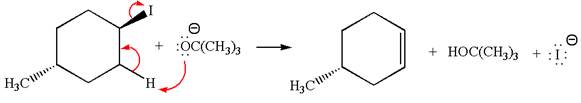
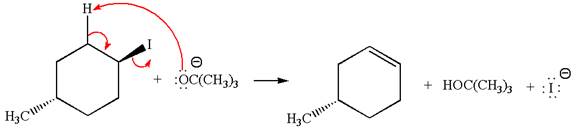
In
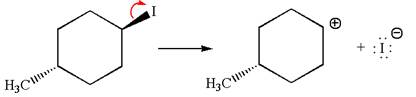
In the second step, the base abstracts the proton from the carbon adjacent to the positively charged carbon. Two products are possible because the proton gets eliminated from two different carbon atoms. The detailed mechanism is shown below:

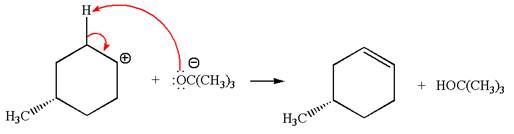
The products formed for the given reaction from both
(d)
Interpretation:
The detailed mechanisms for the given reaction occurring via
Concept introduction:
The
In case of
Answer to Problem 8.44P
The
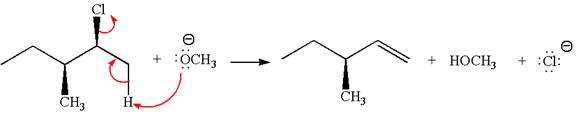
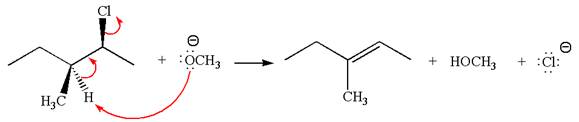
The
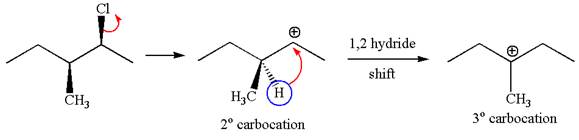
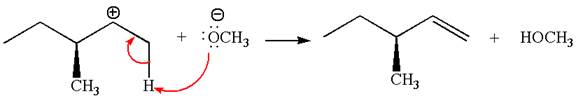
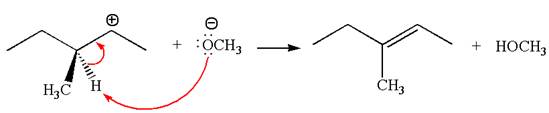
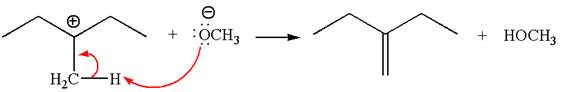
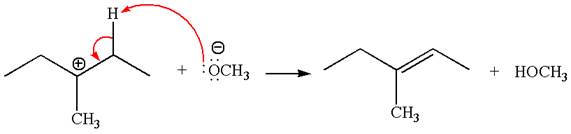
Explanation of Solution
The given reaction equation is:
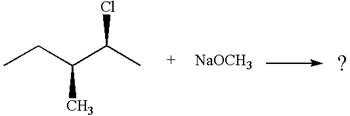
In the given reaction,
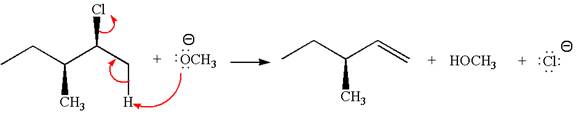
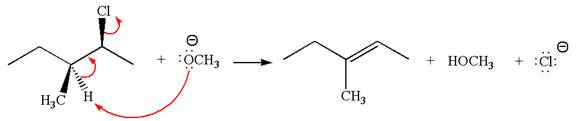
In
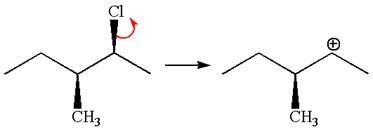
In the second step, the base abstracts the proton from the carbon adjacent to the positively charged carbon. Two products are possible from the secondary carbocation because the proton is eliminated from two different carbon atoms.

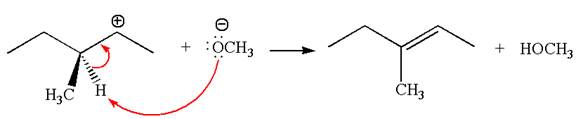
The carbocation formed is a secondary carbocation, which can be rearranged to a more stable tertiary carbocation by
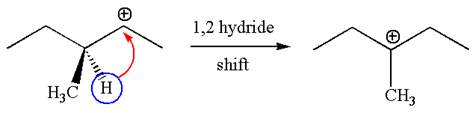
Two products are possible from the tertiary carbocation because the proton is eliminated from two different carbon atoms.
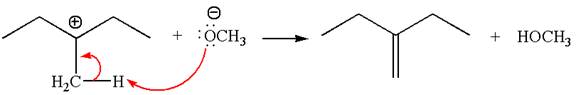
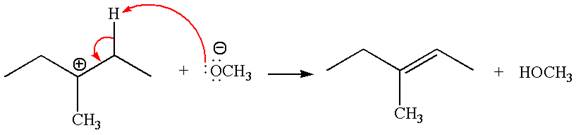
Thus, in all the reactions above, the products are formed by
The products formed for the given reaction from both
(e)
Interpretation:
The detailed mechanisms for the given reaction occurring via
Concept introduction:
The
In case of
Answer to Problem 8.44P
The
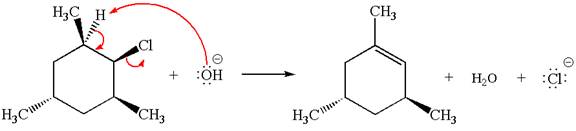
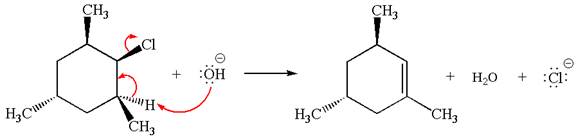
The
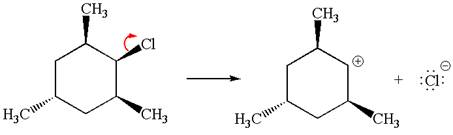
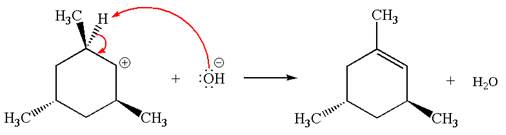
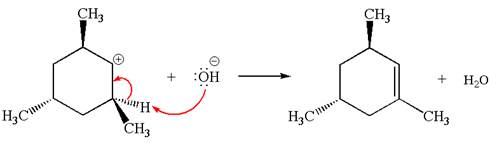
Explanation of Solution
The given reaction equation is:
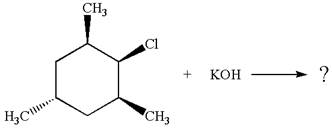
In the given reaction,
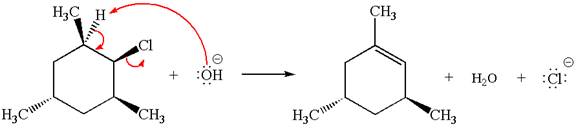

In

In the second step, the base abstracts the proton from the carbon adjacent to the positively charged carbon. Two products are possible because the proton is eliminated from two different carbon atoms. The detailed mechanism is shown below:


The products formed for the given reaction from both
(f)
Interpretation:
The detailed mechanisms for the given reaction occurring via
Concept introduction:
The
In case of
Answer to Problem 8.44P
The
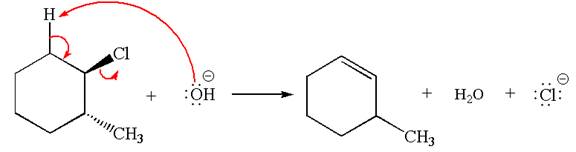
The
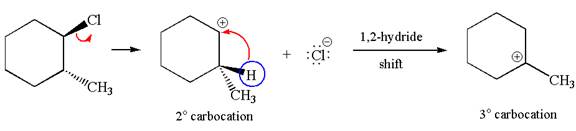
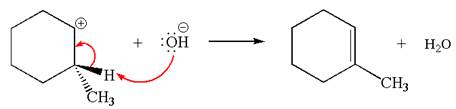
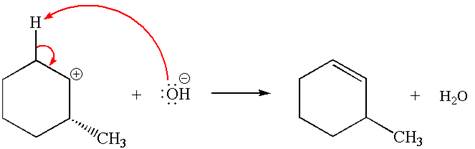
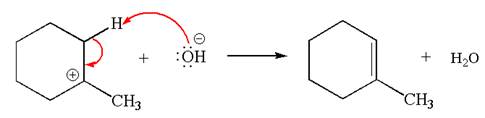
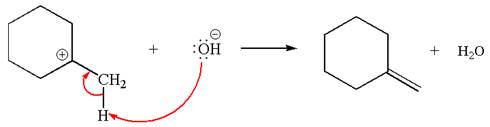
Explanation of Solution
The given reaction equation is:
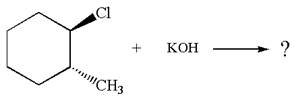
In the given reaction,
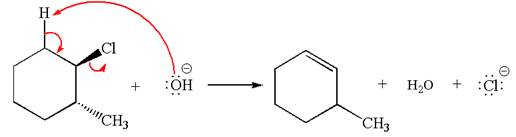
In
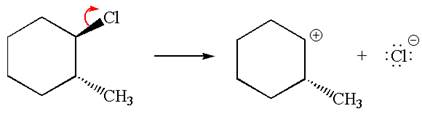
In the second step, the base abstracts the proton from the carbon adjacent to positively charged carbon. Two products are possible because the proton is eliminated from two different carbon atoms. The detailed mechanism is shown below:
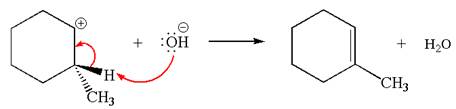
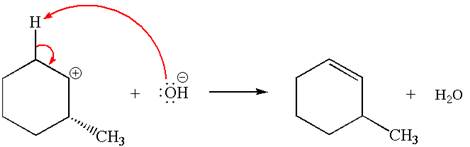
The carbocation formed is a secondary carbocation, which can be rearranged to form more stable tertiary carbocation by
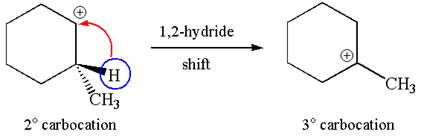
Two products are possible from the tertiary carbocation because the proton IS eliminated from two different carbon atoms.
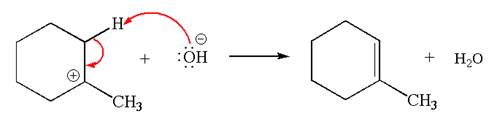
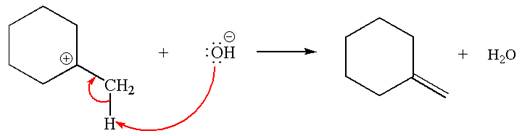
The products formed for the given reaction from both
Want to see more full solutions like this?
Chapter 8 Solutions
EBK GET READY FOR ORGANIC CHEMISTRY
- Nonearrow_forwardDraw the complete mechanism for the following reaction. Predict the product(s) as well. Br₂arrow_forwardDraw a mechanism to account for the formation of the NaOH product in the reaction shown here. Hint: Under these A conditions, deprotonation of a propargylic (C=C-CH) carbon is reversible.arrow_forward
- 3. When the following deuterium-labeled compound is treated with potassium tert-butoxide in DMF, a single product is observed. When the same substrate is heated in the presence of dilute potassium ethoxide in ethanol, a mixture of two products is formed. Provide the complete, detailed mechanism (curved arrows) for each reaction and label each reaction as E1 or E2. Note: deuterium is an isotope of hydrogen and can be treated similarly to hydrogen in chemical reactions but cannot be implied. H KOEt DMF ? D H dilute KOEt EtOH ?+ ?arrow_forwardThe reaction shown here proceeds via a carbocation rearrangement. Draw a complete, detailed mechanism to account for the product. Explain why the carbocation rearrangement is favorable. CH;OH Brarrow_forwardWhen the following deuterium-labeled compound is treated with potassium tert-butoxide in DMF, a single product is observed. When the same substrate is heated in the presence of dilute potassium ethoxide in ethanol, a mixture of two products is formed. Provide the complete, detailed mechanism (curved arrows) for each reaction and label each reaction as E1 or E2. Note: deuterium is an isotope of hydrogen and can be treated similarly to hydrogen in chemical reactions but cannot be implied. D H KO/Bu DMF D H dilute KOET EtOH ?+ ?arrow_forward
- Please draw the mechanism of the reaction shown below.arrow_forwardConsider the following reactants: CI OH Would elimination take place at a significant rate between these reactants? If you said elimination would take place, draw the major products in the upper drawing area. If you said elimination would take place, also draw the complete mechanism for one of the major products in the lower drawing area. If there is more than one major product, you may draw the mechanism that leads to any of them. Oyes Onoarrow_forwardPlease help, correctly with explanation and mechanism need.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
