
EP ORGANIC CHEMISTRY -MOD.MASTERING 18W
9th Edition
ISBN: 9780136781776
Author: Wade
Publisher: PEARSON CO
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8, Problem 8.50SP
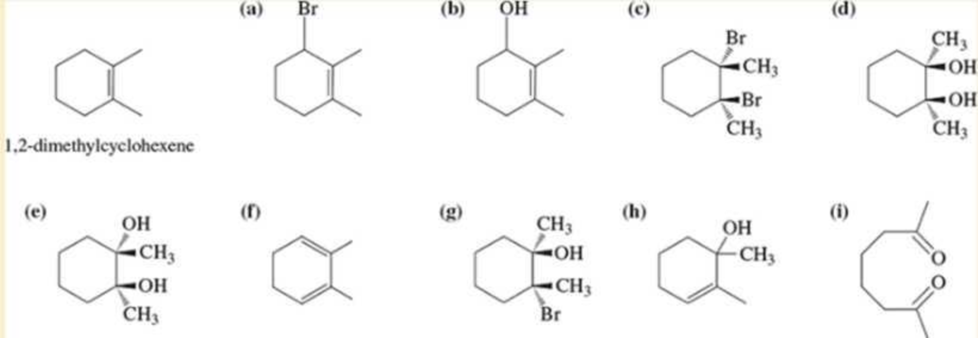
Using 1,2-dimethylcyclohexene as your starting material, show how you would synthesize the following compounds. (Once you have shown how to synthesize a compound, you may use it as the starting material in any later parts of this problem.) If a chiral product is shown, assume that it is part of a racemic mixture.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Using cyclooctyne as your starting material, show how you would synthesize the following compounds. (Once you haveshown how to synthesize a compound, you may use it as the starting material in any later parts of this problem.) ) cyclooctane
Using hex-1-ene as your starting material, show how you would synthesize the following compounds. (Once you haveshown how to synthesize a compound, you may use it as the starting material in any later parts of this problem.) c) 2,2-dibromohexane
Using cyclooctyne as your starting material, show how you would synthesize the following compounds. (Once you haveshown how to synthesize a compound, you may use it as the starting material in any later parts of this problem.) cyclooctanone
Chapter 8 Solutions
EP ORGANIC CHEMISTRY -MOD.MASTERING 18W
Ch. 8.3A - Predict the major products of the following...Ch. 8.3A - a. When 1 mole of buta-1,3-diene reacts with 1...Ch. 8.3B - Predict the major products of the following...Ch. 8.3B - Show how you would accomplish the following...Ch. 8.4B - Propose a mechanism to show how...Ch. 8.4B - Predict the products of the following hydration...Ch. 8.6 - a. Propose a mochansm fortho following reaction....Ch. 8.6 - Prob. 8.8PCh. 8.6 - Prob. 8.9PCh. 8.7A - Prob. 8.10P
Ch. 8.7A - Prob. 8.11PCh. 8.7C - Prob. 8.12PCh. 8.7C - Prob. 8.13PCh. 8.7C - a. When (Z)-3-methylhex-3-ene undergoes...Ch. 8.7C - Prob. 8.15PCh. 8.7C - Prob. 8.16PCh. 8.8B - Prob. 8.17PCh. 8.8B - Prob. 8.18PCh. 8.9 - Prob. 8.19PCh. 8.9 - Prob. 8.20PCh. 8.9 - Prob. 8.21PCh. 8.9 - Prob. 8.22PCh. 8.10 - Prob. 8.23PCh. 8.10 - Prob. 8.24PCh. 8.10 - Prob. 8.25PCh. 8.11A - Prob. 8.26PCh. 8.11B - Prob. 8.27PCh. 8.11B - Prob. 8.28PCh. 8.12 - Prob. 8.29PCh. 8.13 - a. Propose a mechanism for the conversion of...Ch. 8.13 - Magnesium monoperoxyphthalate (MMPP) epoxidizes...Ch. 8.13 - Predict the major products of the following...Ch. 8.13 - When 1,2-epoxycyclohexane (cyclohexene oxide) is...Ch. 8.14C - Predict the major products of the following...Ch. 8.14C - Prob. 8.35PCh. 8.15B - Prob. 8.36PCh. 8.15C - Predict the major products of the following...Ch. 8.16A - Prob. 8.38PCh. 8.16A - Prob. 8.39PCh. 8.16B - Prob. 8.40PCh. 8.16B - Prob. 8.41PCh. 8.16C - Prob. 8.42PCh. 8.17B - Prob. 8.43PCh. 8.17B - Prob. 8.44PCh. 8.17B - Show how you would synthesize each compound,...Ch. 8 - Prob. 8.46SPCh. 8 - Prob. 8.47SPCh. 8 - Give the products expected when the following...Ch. 8 - Show how you would make the following compounds...Ch. 8 - Using 1,2-dimethylcyclohexene as your starting...Ch. 8 - Show how you would synthesize each compound using...Ch. 8 - Prob. 8.52SPCh. 8 - Show how you might use olefin metathesis to...Ch. 8 - Prob. 8.54SPCh. 8 - Prob. 8.55SPCh. 8 - Propose mechanisms consistent with the following...Ch. 8 - Prob. 8.57SPCh. 8 - Prob. 8.58SPCh. 8 - Draw a reaction-energy diagram for the propagation...Ch. 8 - Prob. 8.60SPCh. 8 - Prob. 8.61SPCh. 8 - Prob. 8.62SPCh. 8 - Prob. 8.63SPCh. 8 - Prob. 8.64SPCh. 8 - Prob. 8.65SPCh. 8 - Prob. 8.66SPCh. 8 - Prob. 8.67SPCh. 8 - Prob. 8.68SPCh. 8 - Prob. 8.69SPCh. 8 - Prob. 8.70SPCh. 8 - Prob. 8.71SPCh. 8 - Prob. 8.72SPCh. 8 - Prob. 8.73SPCh. 8 - Prob. 8.74SPCh. 8 - Prob. 8.75SPCh. 8 - Prob. 8.76SPCh. 8 - Prob. 8.77SPCh. 8 - Prob. 8.78SPCh. 8 - Prob. 8.79SP
Additional Science Textbook Solutions
Find more solutions based on key concepts
For each of the following 2-dimensional shapes, determine the highest order rotation axis of symmetry.
Inorganic Chemistry
Practice Exercise 1
Which of the following factors determines the size of an atom? a. the volume of the nucleus...
Chemistry: The Central Science (13th Edition)
Practice Problem ATTEMPT
Write the rate expressions for each of the following reactions:
(a)
(b)
(c)
Chemistry
16.43 The following pictures represent solutions at various stages in thetitration of a weak diprotic acid with...
Chemistry (7th Edition)
Write a Lewis formula for each of the following organic molecules: C2H3Cl (vinyl chloride: starting material fo...
Organic Chemistry - Standalone book
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Show how you would synthesize each compound using methylenecyclohexane asyour starting material. If a chiral product is shown, assume that it is part of a racemicmixture.arrow_forwardUsing hex-1-ene as your starting material, show how you would synthesize the following compounds. (Once you haveshown how to synthesize a compound, you may use it as the starting material in any later parts of this problem.) hex-1-ynearrow_forwardShow how you would convert 2-methylcyclopentanol to the following products. Any of these products may be used as thereactant in any subsequent part of this problem.(a) 1-methylcyclopentenearrow_forward
- Using cyclohexane as your starting material, show how you would synthesize each of the following compounds. (Onceyou have shown how to synthesize a compound, you may use it as the starting material in any later parts of this problem.)(a) bromocyclohexanearrow_forwardUsing cyclooctyne as your starting material, show how you would synthesize the following compounds. (Once you haveshown how to synthesize a compound, you may use it as the starting material in any later parts of this problem.) e) 1,1-dibromocyclooctanearrow_forwardUsing cyclohexane as your starting material, show how you would synthesize each of the following compounds. (Onceyou have shown how to synthesize a compound, you may use it as the starting material in any later parts of this problem.)(a) bromocyclohexane (b) cyclohexene (c) ethoxycyclohexane(d) 3-bromocyclohex-1-ene (e) cyclohexa-1,3-diene (f) cyclohexanolarrow_forward
- Using cyclohexane as your starting material, show how you would synthesize each of the following compounds. (Onceyou have shown how to synthesize a compound, you may use it as the starting material in any later parts of this problem.)(a) bromocyclohexane (b) cyclohexene (c) ethoxycyclohexane(d) 3-bromocyclohex-1-enearrow_forwarda) When (Z)-3-methylhex-3-ene undergoes hydroboration–oxidation, two isomeric products are formed. Give their structures, and label each asymmetric carbon atom as (R) or (S). What is the relationship between these isomers?arrow_forwardUnder certain reaction conditions, 2,3-dibromobutane reacts with two equivalents of base to give three products, each of which contains two new p bonds. Product A has two sp hybridized carbon atoms, product B has one sp hybridized carbon atom, and product C has none. What are the structures of A, B, and C?arrow_forward
- Predict the major product of the following reaction. If a single enantiomer will be obtained, be sure to draw it with well-defined stereochemistry. If a mixture of enantiomers will be obtained, either draw both enantiomers or label the product as "racemic".arrow_forwardNonconjugated , -unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium with their conjugated , -unsaturated isomers. Propose a mechanism for this isomerization.arrow_forwardIf phenoxide ion is allowed to react with 1-bromopentane, pentyl phenyl ether is obtained. However, if cyclohexane is used as the alkyl halide, the major products are phenol and cyclohexene. Explain how these products were formed.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
How to Design a Total Synthesis; Author: Chemistry Unleashed;https://www.youtube.com/watch?v=9jRfAJJO7mM;License: Standard YouTube License, CC-BY