
Concept explainers
(a)
Interpretation:
Whether a carbocation rearrangement will take place in an
Concept introduction:
The first step in an
A carbocation may undergo rearrangement through a
Stability of carbocations increases as
Answer to Problem 8.52P
A carbocation rearrangement will take place for this substrate.
The rearrangement can be drawn using curved arrow notation as
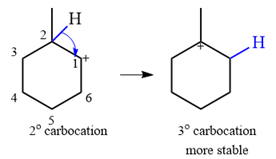
Explanation of Solution
The given substrate initially forms the carbocation shown below in an
It is a relatively stable secondary carbocation. It will undergo a rearrangement only if it leads to the formation of a more stable tertiary carbocation. Two
Therefore, a carbocation rearrangement is possible in this case.
The rearrangement can be drawn using the curved arrow notation as
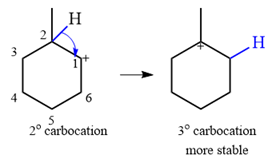
Formation of a more stable tertiary carbocation results in the rearrangement for this substrate.
(b)
Interpretation:
Whether a carbocation rearrangement will take place in an
Concept introduction:
The first step in an
A carbocation may undergo rearrangement through a
Stability of carbocations increases as
Answer to Problem 8.52P
There will be no carbocation rearrangement in the case of this substrate.
Explanation of Solution
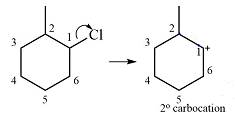
The substrate and the carbocation that it will be formed initially in an
The carbocation initially formed is a relatively stable secondary carbocation. There are two hydrogen atoms on adjacent carbons (C2 and C6) that can undergo a
Therefore, a carbocation rearrangement will not take place in this case.
There is no carbocation rearrangement for this case as there is no gain in stability.
(c)
Interpretation:
Whether a carbocation rearrangement will take place in an
Concept introduction:
The first step in an
A carbocation may undergo rearrangement through a
Stability of carbocations increases as
Answer to Problem 8.52P
A carbocation rearrangement will not take place in this case.
Explanation of Solution
The given substrate initially forms the carbocation shown below in an
The carbocation formed is a tertiary carbocation, the most stable one. Therefore, a rearrangement will occur only if it leads to another tertiary carbocation that is further stabilized by resonance. No resonance stabilization is possible here as there are no double bonds in the carbocation.
Therefore, a carbocation rearrangement will not take place in this case.
Carbocation rearrangement is not possible because the one initially formed is a tertiary carbocation.
(d)
Interpretation:
Whether a carbocation rearrangement will take place in an
Concept introduction:
The first step in an
A carbocation may undergo rearrangement through a
Stability of carbocations increases as
Answer to Problem 8.52P
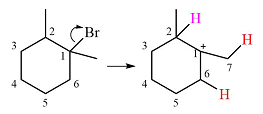
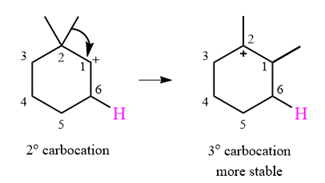
A carbocation rearrangement will take place in this case.
The curved arrow notation for the rearrangement can be drawn as
Explanation of Solution
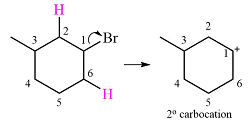
The substrate shown and the carbocation that it will initially form in this case is
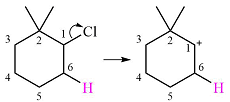
The carbocation initially formed is a relatively stable secondary carbocation. It will undergo a rearrangement only if it leads to the formation of a resonance stabilized secondary carbocation or a tertiary carbocation. Only one hydride shift is possible, but it will not occur as the carbocation formed will be a similar secondary carbocation.
A methyl shift, from C2 to C1 will, however, lead to the formation of a tertiary carbocation.
Therefore, a carbocation rearrangement will take place in this case.
The curved arrow notation for this rearrangement can be drawn as
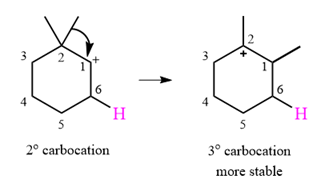
A
(e)
Interpretation:
Whether a carbocation rearrangement will take place in an
Concept introduction:
The first step in an
A carbocation may undergo rearrangement through a
Stability of carbocations increases as
Answer to Problem 8.52P
A carbocation rearrangement will take place in this case.
The curved arrow notation for this rearrangement is
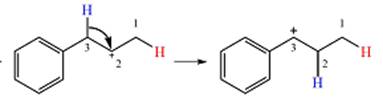
Explanation of Solution
The given substrate initially forms the carbocation shown below in an
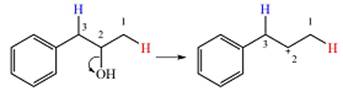
Two
The other, from C3 to C2 will result in another secondary carbocation. This would normally not lead to a more stable carbocation. However, in this case, the charge is now in a conjugated position with the
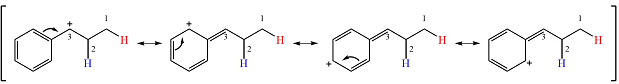
Therefore, a rearrangement will take place for this substrate.
The curved arrow notation for the rearrangement can be drawn as
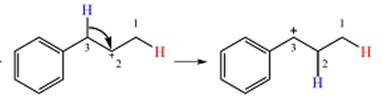
Resonance can increase the stability of a carbocation as the number of atoms on which the charge is delocalized increases.
(f)
Interpretation:
Whether a carbocation rearrangement will take place in an
Concept introduction:
The first step in an
A carbocation may undergo rearrangement through a
Stability of carbocations increases as
Answer to Problem 8.52P
A carbocation rearrangement will take place in this case.
The curved arrow notation for the rearrangement can be drawn as
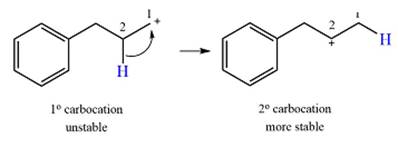
Explanation of Solution
The given substrate initially forms the carbocation, as shown below, in an
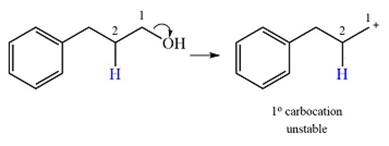
The carbocation initially formed is an unstable primary carbocation. Any rearrangement that converts it to a secondary or a tertiary carbocation will be favorable. There is only one possible
Therefore, a rearrangement of the carbocation will take place for this substrate.
The curved arrow notation for the rearrangement can be drawn as

A primary carbocation is unstable and will rearrange to a secondary carbocation where possible.
(g)
Interpretation:
Whether a carbocation rearrangement will take place in an
Concept introduction:
The first step in an
A carbocation may undergo rearrangement through a
Stability of carbocations increases as
Answer to Problem 8.52P
A carbocation rearrangement will take place in this case.
A curved arrow notation for the rearrangement can be drawn as
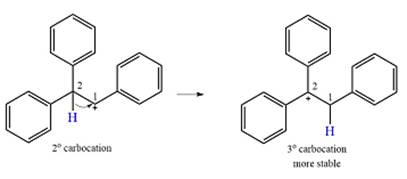
Explanation of Solution
The substrate and the carbocation that will initially be formed in an
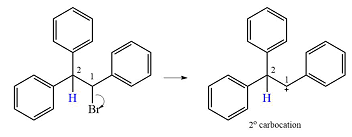
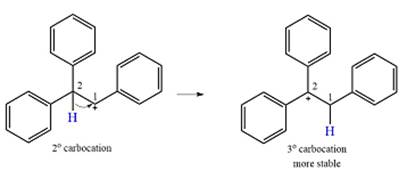
The carbocation initially formed is a relatively stable secondary carbocation. The presence of a benzene ring on the same carbon will lead to further stabilization by resonance. Resonance will delocalize the charge over a total of four carbon atoms.
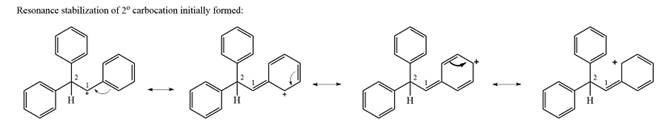
A
The much higher stability of this carbocation will mean a carbocation rearrangement will take place for this substrate.
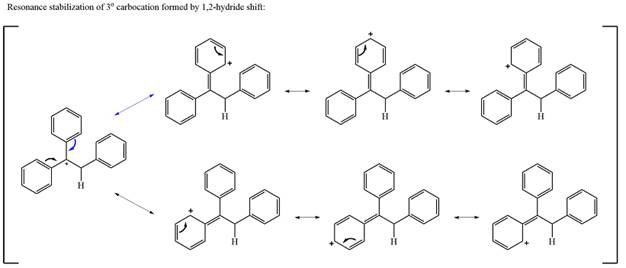
The curved arrow notation for this rearrangement can be drawn as
Formation of a more stable tertiary carbocation leads to the rearrangement.
(h)
Interpretation:
Whether a carbocation rearrangement will take place in an
Concept introduction:
The first step in an
A carbocation may undergo rearrangement through a
Stability of carbocations increases as
Answer to Problem 8.52P
No carbocation rearrangement will take place in this case.
Explanation of Solution
The given substrate and the carbocation that it forms initially in an
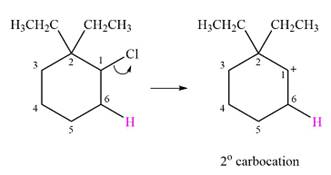
The carbocation formed is a relatively stable secondary carbocation. There is only one possible rearrangement, a
Therefore, a carbocation rearrangement will not take place in this case.
A carbocation rearrangement will not take place if it does not result in a more stable carbocation.
(i)
Interpretation:
Whether a carbocation rearrangement will take place in an
Concept introduction:
The first step in an
A carbocation may undergo rearrangement through a
Stability of carbocations increases as
Answer to Problem 8.52P
A carbocation rearrangement will take place in this case.
The curved arrow representation of this rearrangement can be drawn as
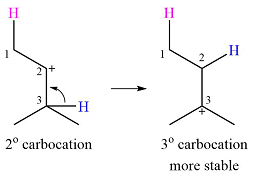
Explanation of Solution
The given substrate and the carbocation it will form in an
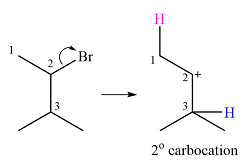
There are two possible
The second one, from C3 to C2, will result in a more stable tertiary carbocation.
Therefore, a carbocation rearrangement is possible in this case.
The curved arrow representation of this rearrangement can be drawn as

The carbocation rearrangement is possible because a more a stable carbocation is formed.
Want to see more full solutions like this?
Chapter 8 Solutions
ORGANIC CHEMISTRY PRINCIPLES & MECHANISM
- Solve it asaparrow_forwardHello, I am very confused working on this worksheet, please help me understand every step for either reacton or both that I have photos uploaded of!arrow_forwardConsider the molecule given below. In theory, there are only two inequivalent hydrogens in this molecule that could be substituted by Br in a free radical bromination – circle them. Put an asterisk to mark the one most likely to be substituted first. However, there are 5 possible products from free radical bromination. Draw all the products and show using arrow formalism how the intermediate radicals leading to these products formedarrow_forward
- Please draw a clear imagearrow_forward5. Provide the product that will result for the following reaction and provide the complete mechanism. CH,N2 НО 오arrow_forward. Use to curved arrow notation, propose a mechanism for the following reaction and state whether it is either SN1, E1, SN2, or E2. Give the IUPAC names of all organic reactants and products.arrow_forward
- Give mechanism (name) for the reaction belowarrow_forwardUsing curved-arrow notation, write out the mechanism for the following halogenation reaction. Be sure to label steps as initiation, propagation and termination. Only one termination step is needed. Br₂, light Brarrow_forwardDraw the first step in the mechanism for the acid-catalyzed addition of HCI to the alkene in the drawing area below. Be sure you draw only the first step. It's OK if the product of this step is a short-lived intermediate. • You can add any necessary small molecule reactants on the reactant side of the step. • You can leave out any small molecule byproducts on the product side of the step. • Be sure to draw in any lone pairs that are moved by curved arrows. : ☐ ↑ Click and drag to start drawing a structure.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
