
Concept explainers
(a)
Interpretation:
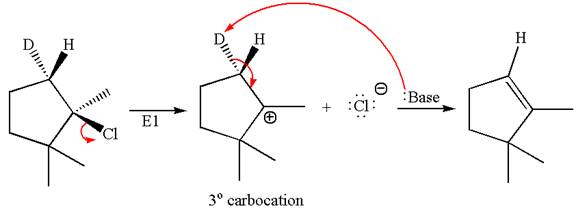
The detailed mechanism for the given reaction occurring via
Concept introduction:
The deuterium
Answer to Problem 8.64P
The
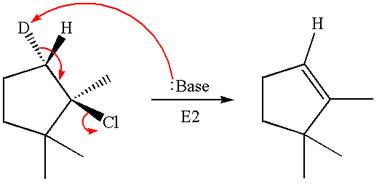
Explanation of Solution
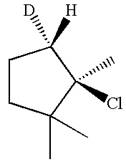
The given reaction equation is
Here, the chlorine atom is the leaving group, which can be eliminated along with either
In the given substrate, the leaving group
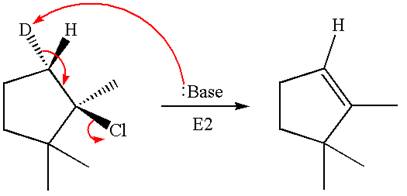
The product formed for the given reaction from
(b)
Interpretation:
The detailed mechanism for the given reaction occurring via
Concept introduction:
The deuterium
Answer to Problem 8.64P
The
Elimination of
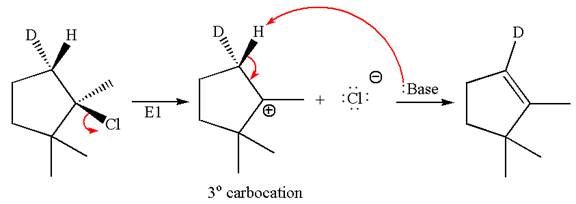
Elimination of
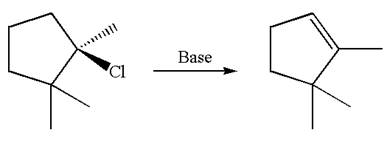
Explanation of Solution
The given reaction equation is
Here, the chlorine atom is the leaving group, which can be eliminated along with either
According to
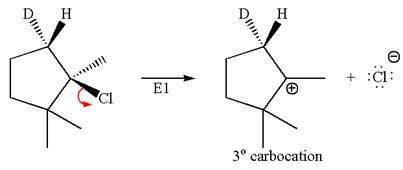
In the second step, both
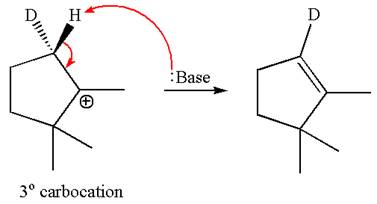
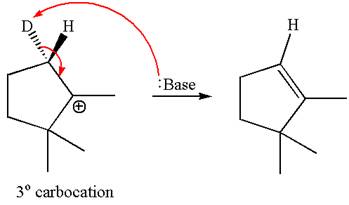
The product formed for the given reaction from
(c)
Interpretation:
The molar mass of each product from
Concept introduction:
The molar mass is the sum of the
The
Answer to Problem 8.64P
The molar masses of the products formed by
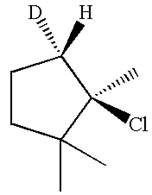
Explanation of Solution
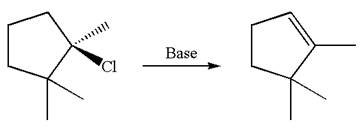
The given reaction equation is
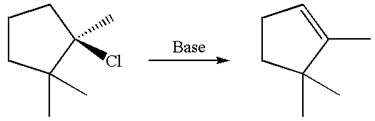
Here, the chlorine atom is the leaving group, which can be eliminated along with either
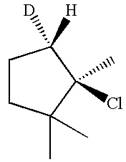
In the given substrate, the leaving group
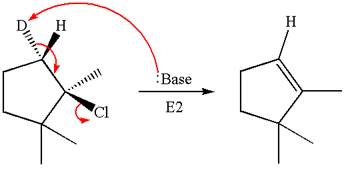
The molecular formula for this product is
According to
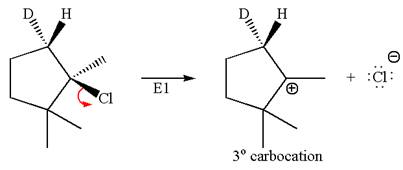
In the second step, both
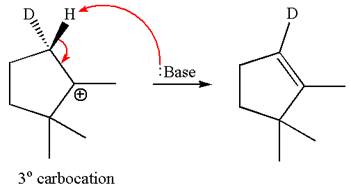
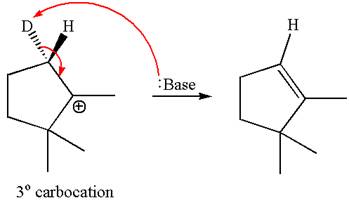
The molecular formulae for both the products formed by
Hence the molar mass of the product
The molar masses of each product formed by
Want to see more full solutions like this?
Chapter 8 Solutions
EBK GET READY FOR ORGANIC CHEMISTRY
- Write the rate law for the reaction in Problemarrow_forwardHeterolysis of the C–Z bond can generate a carbocation or a carbanion. Explain How ?arrow_forwardDraw the mechanism and the energy diagram for the reaction shown below. Include any resonance structures for the intermediates of the reaction. H3O+arrow_forward
- Show the mechanism of this reaction step by steparrow_forwardThe following reaction, which is discussed in Chapter 8, is an example of a unimolecular nucleophilic substitution (Sn1) reaction. It consists of the four elementary steps shown here. For each step (i-iv), (a) identify all electron-rich sites and all electron-poor sites, (b) draw in the appropriate curved arrows to show the bond formation and bond breaking that occur, and (c) name the elementary step. (i) H-OCH3 OH + H-OCH3 H. H (ii) ©CH2 + H,O (iii) ©CH2 CH3 + HO-CH3 (iv) CH3 CH3 + H2O-CH, + НО—СНЗ foarrow_forwardThe reaction shown in the box forms only one product that exists as a racemic mixture. The other possible product – shown outside the box – is NOT observed. Briefly explain in words why only the observed product is formed. Hint: consider the mechanism. Br HBr OCH3 OCH3 OCH3 Br racemic mixture pdt not observedarrow_forward
- The following reaction, which is discussed in Chapter 17, consists of the two elementary steps shown. For each step (i and ii), (a) identify all electron-rich sites and all electron-poor sites, (b) draw in the appropriate curved arrows to show the bond formation and bond breaking that occur, and (c) name the elementary step. (i) (C6H5)3P + + BrO H Br H P(C6H5)3 (ii) (CH),CO + (CH3)3CH H P(C6H5)3arrow_forwardDraw the first step in the mechanism for the acid-catalyzed addition of HCI to the alkene in the drawing area below. Be sure you draw only the first step. It's OK if the product of this step is a short-lived intermediate. • You can add any necessary small molecule reactants on the reactant side of the step. • You can leave out any small molecule byproducts on the product side of the step. • Be sure to draw in any lone pairs that are moved by curved arrows. : ☐ ↑ Click and drag to start drawing a structure.arrow_forwardDraw the most stable resonance form for the intermediate in the following electrophilic substitution reaction. LOCH3 LOCH3 HNO3 / CH3CO₂H O₂N • You do not have to consider stereochemistry. • Include all valence lone pairs in your answer. ● In cases where there is more than one answer, just draw one.arrow_forward
- Consider the mechanism of the reaction shown below. Give the structure of the next important organic reaction intermediate along the reaction coordinate. Your answer could be the fina HBr Edit Click on the drawing box above to activate the MarvinSketch drawing tool and then draw your answer to this question. If there is no reaction, then check the "no reaction" box below. no reactionarrow_forwardWhat is/are the product(s) of the reaction of 2-bromopropane with NaOCOCH3? Draw the mechanism & identify it as Sn1, Sn2, E1, or E2. Please explain your reasoning.arrow_forwardConsider the following reactants: CI OH Would elimination take place at a significant rate between these reactants? If you said elimination would take place, draw the major products in the upper drawing area. If you said elimination would take place, also draw the complete mechanism for one of the major products in the lower drawing area. If there is more than one major product, you may draw the mechanism that leads to any of them. Oyes Onoarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
