
Concept explainers
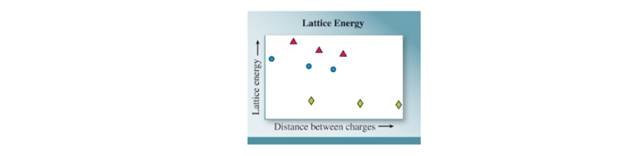
Lattice energies are graphed for three series of compounds in which the ion charges are
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
Connect 1-semester Access Card For Chemistry
- 1. Given the following information, calculate the lattice energy for LiI(s). Show all work and construct a diagram(cycle) that shows each step in the process of forming LiI(s) from gaseous ions.arrow_forwardTrue or false? In general, a large atom has a smaller electro-negativity. Explain.arrow_forwardThe lattice energy of MgO is 3890 kJ/mol. The first and the second ionization energies (IE1 and IE2) of Mg are 738 kJ/mol and 1450.6 kJ/mol, respectively. The first ionization energy of O is 1314 kJ/mol. The first electron affinity (EA1) of O is +141 kJ/mol. Using these data, as well as data from a table of thermodynamic data at 1 atm and 25°C, determine the second electron affinity for oxygen, EA2(O). kJ/molarrow_forward
- Construct a Born–Haber cycle for the formation of the hypotheticalcompound NaCl2, where the sodium ion has a 2+charge (the second ionization energy for sodium is given inTable 7.2). (a) How large would the lattice energy need to befor the formation of NaCl2 to be exothermic? (b) If we wereto estimate the lattice energy of NaCl2 to be roughly equalto that of MgCl2 (2326 kJ/mol from Table 8.1), what valuewould you obtain for the standard enthalpy of formation,ΔHf°, of NaCl2?arrow_forwardEnergy is required to remove two electrons from Ca to formCa2+, and energy is required to add two electrons to O toform O2 - . Yet CaO is stable relative to the free elements.Which statement is the best explanation? (a) The latticeenergy of CaO is large enough to overcome these processes.(b) CaO is a covalent compound, and these processes areirrelevant. (c) CaO has a higher molar mass than either Ca orO. (d) The enthalpy of formation of CaO is small. (e) CaO isstable to atmospheric conditions.arrow_forwardThe compound gallium arsenide is a commonly used semiconductor,having an energy gap Eg of 1.43 eV. Its crystal structureis like that of silicon, except that half the silicon atoms are replacedby gallium atoms and half by arsenic atoms. Draw a flattened-outsketch of the gallium arsenide lattice, following the pattern.What is the net charge of the (a) gallium and (b) arsenic ioncore? (c) How many electrons per bond are there? (Hint: Consultthe periodic table in Appendix G.)arrow_forward
- Aluminum oxide (Al₂ O₃) is a widely used industrial abrasive(emery, corundum), for which the specific application depends onthe hardness of the crystal. What does this hardness imply about the magnitude of the lattice energy? Would you have predictedfrom the chemical formula that Al₂ O₃ is hard? Explain.arrow_forwardNaCl and KF have the same crystal structure. The only differencebetween the two is the distance that separates cationsand anions. (a) The lattice energies of NaCl and KF are givenin Table 8.1. Based on the lattice energies, would you expectthe Na¬Cl or the K¬F distance to be longer? (b) Use theionic radii given in Figure 7.8 to estimate the Na¬Cl andK¬F distances.arrow_forward(a) Based on the lattice energies of MgCl2 and SrCl2 given inTable 8.1, what is the range of values that you would expectfor the lattice energy of CaCl2? (b) Using data from AppendixC, Figure 7.11, Figure 7.13, and the value of the secondionization energy for Ca, 1145 kJ>mol, calculate the latticeenergy of CaCl2.arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





