
(a)
Interpretation:- The products formed by assigning the appropriate mass spectrum to each compound needs to be confirmed. The parent and the base peaks needs to be specified.
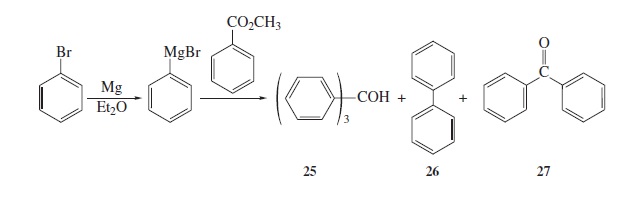
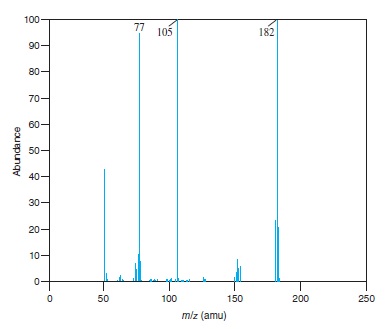
Fig − 8.61
Mass spectrum of component of reaction mixture
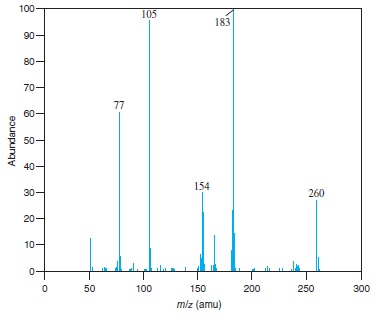
Fig − 8.62
Mass spectrum of component of reaction mixture
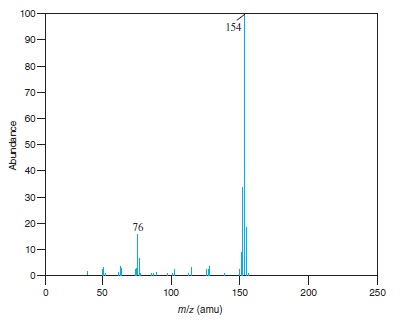
Fig − 8.63
Mass spectrum of component of reaction mixture
Concept Introduction: Fragmentation is the process in which energetically unstable molecular ions gets dissociated, when these molecules are passed through the ionization chamber of a mass spectrometer.
(b)
Interpretation:- The compound represented by the mass spectrum needs to be identified.
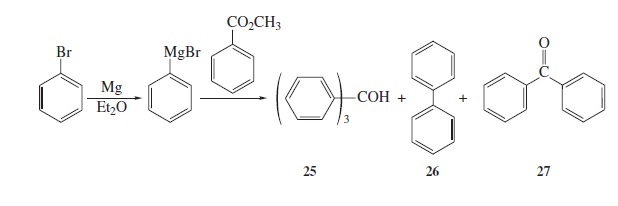
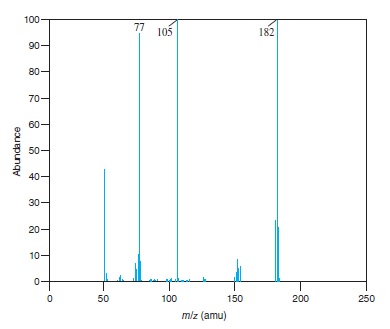
Fig − 8.61
Mass spectrum of component of reaction mixture
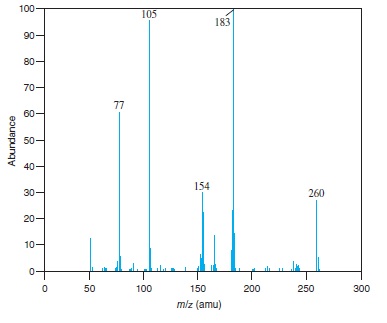
Fig − 8.62
Mass spectrum of component of reaction mixture
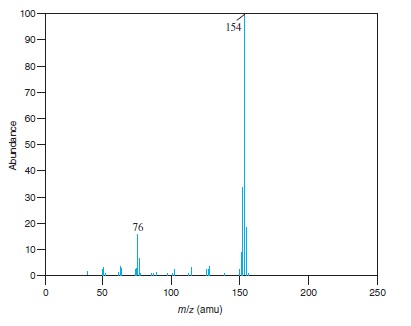
Fig − 8.63
Mass spectrum of component of reaction mixture
Concept Introduction: Fragmentation is the process in which energetically unstable molecular ions gets dissociated, when these molecules are passed through the ionization chamber of a mass spectrometer.
(c)
Interpretation: The structure of the ion for each of the peaks for which value of
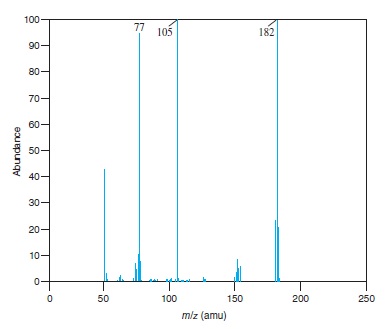
Fig − 8.61
Mass spectrum of component of reaction mixture
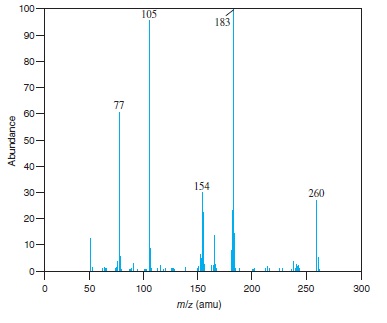
Fig − 8.62
Mass spectrum of component of reaction mixture
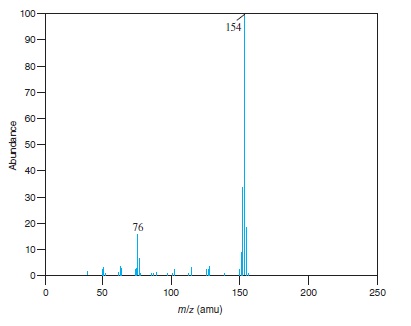
Fig − 8.63
Mass spectrum of component of reaction mixture
Concept Introduction: Fragmentation is the process in which energetically unstable molecular ions gets dissociated, when these molecules are passed through the ionization chamber of a mass spectrometer.
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
- Explain the differences in acidity between p-cyanobenzoic acid and m-cyanobenzoic acid. Illustrate your answer appropriately, showing the acid-base reaction that takes place with each of these and explaining in such terms as electronic (inductive or resonance) and/or steric effects.arrow_forwardAccount for the greater amount of internal pressure in the aspirin extraction versus the ß- naphthol extraction.Why does the sequence for extracting the diethyl ether solution of aspirin acid, naphthalene, and ß-naphthol start with aqueous bicarbonate and follow with aqueous hydroxide rather than the reverse order?arrow_forwardWhy is the refluxing time required for the synthesis of methyl salicylate is quite long?arrow_forward
- Propose an experimental design on the separation of components of the mixture of o-nitrophenol and p-nitrophenol., provide a schematic diagram of the step-by-step procedurearrow_forwardIn addition to the normal dangers in using concentrated hydrochloric acid, what additional dangers are presented in mixing it with ice and what precautions should be taken? What gas is evolved when hydrochloric acid is added to the borohydride reduction with benzophenone?arrow_forward9-Aminofluorene has applications in the structural analysis of proteins and carbohydrates. Write a stepwise procedure with equations to show how to separate a mixture of 9-aminofluorene and fluorene in diethyl ether solution.arrow_forward
- TLC, a powerful analytical tool, can be used to monitor the progress of reactions. The synthesis of ethyl-3-coumarincarboxylate can be monitored by TLC by displaying the starting aldehyde 1, and coumarin product 2, which have very distinct Rf values (Hint: Think about the polarity of compounds 1 and 2 in terms of their abilities to H-bonds to silica gel). What can be determine about the progress of the reaction from analysis of the TLC shown below?arrow_forwardIn preparation of p-Nitroaniline, Starting material is 50g of p-nitroacetanilide, 22 mL of HCI, 20g of p-nitroaniline. Give % yield of nitro acetanilide. What is the objective of experiment?arrow_forwardWrite the mechanism for the Friedel Crafts Acylation reaction of ethylbenzene using acetyl chloride and aluminum chloride. What ratio of ortho-, para-, and meta- products would you expect? Explain why you expect that ratio.arrow_forward
- Sodium hydroxide was used to help separate benzoic acid and fluorenone because deprotonated benzoic acid (sodium benzoate) favorably partitions into the aqueous phase (and fluorenone does not). Would this procedure have worked without the sodium hydroxide (using simply neutral water)? Why or why not?arrow_forwardWrite a balanced reaction and mechanism for the acid-base reaction of diethylbenzyl phosphonate with sodium hydroxide. Which Hydrogens are most acidic? Explain by writing resonance structures.arrow_forwardWhy phenolphthalein cannot show any colour in pure water media?arrow_forward
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
