
a) Aqueous acidic KMnO4
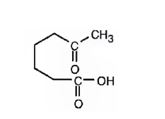
Interpretation:
The product expected when 1-methylcyclohexene reacts with aqueous acidic KMnO4 is to be stated.
Concept introduction:
Potassium permanganate in neutral or aqueous acidic solutions cleaves the double bonds in
To state:
The product expected when 1-methylcyclohexene reacts with aqueous acidic KMnO4.
b)
Interpretation:
The product expected when 1-methylcyclohexene reacts with O3, followed by Zn, CH3COOH is to be stated.
Concept introduction:
Ozone adds to the double bond in alkenes to produce compounds called ozonides. The ozonides upon immediate treatment with Zn and acetic acid yield carbonyl compounds. Each carbon in the double bond cleaved gets attached to an oxygen atom. A di-substituted carbon in double bond gives a ketone while a mono-substituted carbon in double bond gives a ketone and an
To state:
The product expected when 1-methylcyclohexene reacts with O3, followed by Zn, CH3COOH.
Trending nowThis is a popular solution!

Chapter 8 Solutions
Organic Chemistry
- A step in a synthesis of PGE1 (prostaglandin E1, alprostadil) is the reaction of a trisubstituted cyclohexene with bromine to form a bromolactone. Propose a mechanism for formation of this bromolactone and account for the observed stereochemistry of each substituent on the cyclohexane ring. Alprostadil is used as a temporary therapy for infants born with congenital heart defects that restrict pulmonary blood flow. It brings about dilation of the ductus arteriosus, which in turn increases blood flow in the lungs and blood oxygenation.arrow_forwardReaction of iodoethane with CN- yields a small amount of isonitrile, CH3CH2N≡C, along with the nitrile CH3CH2N≡N, as the major product. Write electron-dot structures for both products, assign formal charges as necessary, and propose mechanisms to account for their formation.arrow_forwardBicyclo-2,5-heptadiene can be prepared in two steps from cyclopentadiene and vinyl chloride. Provide a mechanism for each step.arrow_forward
- Compound A (C7H11Br) is treated with magnesium in ether to give B (C7H11MgBr), which reacts violently with D2O to give 1-methylcyclohexene with a deuterium atom on the methyl group (C). Reaction of B with acetone (CH3COCH3) followed by hydrolysis gives D (C10H18O). Heating D with concentrated H2 SO4 gives E (C10 H16), which decolorizes two equivalents of Br2 to give F (C10H16 Br4). E undergoes hydrogenation with excess H2 and a Pt catalyst to give isobutylcyclohexane. Determine the structures of compounds A through F, and show your reasoning throughout.arrow_forwardCompound A (C7H11Br) is treated with magnesium in ether to give B (C7H11MgBr) which reacts violently with D2O to give 1-methylcyclohexene with a deuterium atom on the methyl group (C). Reaction of B with acetone (Ch3COCH3) followed by hydrolysis gives D (C10H18O). Heating D with concentrated H2SO4 gives E (C10H16), which reacts with 2 equivalents of Br2 to give F (C10H16Br4). E undergoes hydrogenation with excess H2 and Pt catalyst to give 2-methylpropylcyclohexane. Determine the structures of compounds A through F, and show your reasoning throughout.arrow_forward1. Deduce the structure of each compound from the information given. All unknowns in this problem have molecular formula C8H12. (a) Upon catalytic hydrogenation, unknown W gives cyclooctane. Ozonolysis of W, followed by reduction with dimethylsulfide, gives octanedioic acid, HOOC—(CH2)6—COOH. Draw the structure of W. (b) Upon catalytic hydrogenation, unknown X gives cyclooctane. Ozonolysis of X, followed by reduction with dimethyl sulfide, gives two equivalents of butanedial, O=CH—CH2CH2—CH=O. Draw the structure of X.arrow_forward
- Compound A has the formula C9H19Cl. B is a C9H19Br compound.A and B undergo base-promoted E2 elimination to give the same alkene C as the major product as well as different minor products.C reacts with one molar equivalent of hydrogen in the presence of a palladium catalyst to form 2,6-dimethylheptane.Addition of HCl to C yields A as the major product.Propose structures for A and B.arrow_forward2-bromopentane, when treated with alcoholic KOH yields a mixture of three alkenes A, B and C. Identify A, B and C. Which is predominant?arrow_forwardCompound A (C7H11Br) is treated with magnesium in ether to give B (C7H11MgBr), which reacts violently with D2O togive 1-methylcyclohexene with a deuterium atom on the methyl group (C). Reaction of B with acetone (CH3COCH3)followed by hydrolysis gives D (C10H18O). Heating D with concentrated H2SO4 gives E (C10H16), which decolorizestwo equivalents of Br2 to give F (C10H16Br4). E undergoes hydrogenation with excess H2 and a Pt catalyst to giveisobutylcyclohexane. Determine the structures of compounds A through F, and show your reasoning throughoutarrow_forward
- What is the major product formed from reacting cyclohexene in aqueous Br2, followed by treatment with potassium tert-butoxide?arrow_forwardA certain hydrocarbon, C5H6, yields the two products shown below upon ozonolysis with 1. O3/CH2Cl2 2. Zn/H3O . Deduce the structure of the hydrocarbon.arrow_forwardWhat alkene would yield 2,2-dimethoxycyclopentane-1,3-dicarbaldehyde on treatment with O3 followed by (CH3)2S?arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

