
ORGANIC CHEMISTRY (LL) W/ACCESS
4th Edition
ISBN: 9781119856122
Author: Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Question
Chapter 8.9, Problem 23ATS
Interpretation Introduction
Interpretation: The products of the catalytic hydrogenation of compound 1 with stereoisomers should be determined.
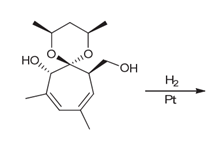
Concept introduction:
Some examples of addition reactions of
The catalytic hydrogenation of alkene leads to the formation of saturated hydrocarbons that are
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The sex attractant by which the female housefly attracts the male has the molecular formula C23H46. Catalytic hydrogenation yields an alkane of molecular formula C23H48. Ozonolysis yieldsWhat is the structure of the housefly sex attractant?
Compound A has molecular formula C7H15B.. Treatment of compound A with sodium ethoxide yields only one elimination product
(compound B) and no substitution products. When compound B is treated with dilute sulfuric acid, compound C is obtained, which
has molecular formula C7H160. Draw the structures of compounds A, B, and C.
Compound A exhibits a peak in its 1H NMR spectrum at 7.6 ppm, indicating that it is aromatic. (a) How are the carbon atoms of the triple bonds hybridized? (b) In what type of orbitals are the π electrons of the triple bonds contained? (c) How many π electrons are delocalized around the ring in A?
Chapter 8 Solutions
ORGANIC CHEMISTRY (LL) W/ACCESS
Ch. 8.3 - Provide a systematic name for each of the...Ch. 8.3 - Prob. 2CCCh. 8.3 - Prob. 3CCCh. 8.3 - Prob. 4CCCh. 8.5 - Prob. 5CCCh. 8.5 - Prob. 6CCCh. 8.5 - Prob. 1LTSCh. 8.5 - Prob. 7PTSCh. 8.5 - Prob. 8ATSCh. 8.5 - Prob. 9CC
Ch. 8.5 - Prob. 2LTSCh. 8.5 - Prob. 10PTSCh. 8.5 - Prob. 11ATSCh. 8.6 - Prob. 12CCCh. 8.6 - Prob. 13CCCh. 8.6 - Prob. 3LTSCh. 8.6 - Prob. 14PTSCh. 8.6 - Prob. 15ATSCh. 8.7 - Predict the product for each reaction, and predict...Ch. 8.7 - Prob. 17CCCh. 8.8 - Prob. 18CCCh. 8.8 - Prob. 19CCCh. 8.8 - Prob. 4LTSCh. 8.8 - Prob. 20PTSCh. 8.8 - Prob. 21ATSCh. 8.9 - Prob. 5LTSCh. 8.9 - Prob. 22PTSCh. 8.9 - Prob. 23ATSCh. 8.10 - Prob. 24CCCh. 8.10 - Prob. 6LTSCh. 8.10 - Prob. 25PTSCh. 8.10 - Prob. 26ATSCh. 8.10 - Prob. 27ATSCh. 8.11 - Prob. 7LTSCh. 8 - Prob. 47PP
Knowledge Booster
Similar questions
- An unknown compound A (molecular formula C7H14O) was treated withNaBH4 in CH3OH to form compound B (molecular formula C7H16O).Compound A has a strong absorption in its IR spectrum at 1716 cm−1.Compound B has a strong absorption in its IR spectrum at 3600−3200cm−1. The 1H NMR spectra of A and B are given. What are the structuresof A and B?arrow_forwardCyclopropenones are described as having aromatic character. How would you account for this, given that the ring contains three ℼ-electronarrow_forwardWrite the structures of all the constitutionally isomeric ethers of molecular formula C5H12O, and give an acceptable name for each.arrow_forward
- An optically active unknown compound B, whose molecular formula is C6H10, reacts with H2/Ni produce compound C, whose molecular formula is C6H14. Compound C is optically inactive. What are the structures of compound B and C ?arrow_forwardCompounds A and B are isomers having molecular formula C5H12. Heating A with Cl2 gives a single product of monohalogenation, whereas heating B under the same conditions forms three constitutional isomers. What are thestructures of A and B?arrow_forwardThere are three constitutional isomers with the molecular formula C5H12. Chlorination of one of these isomers yields only one product. Identify the isomer:arrow_forward
- Compound X (molecular formula C10H12O) was treated with NH2NH2, -OH to yield compound Y (molecular formula C10H14). Based on the 1H NMR spectra of X and Y given below, what are the structures of X and Y?arrow_forwardA compound has the molecular formula C6H12O2. Its IR spectrum shows a strong absorption band near 1740 cm-1. Its 1H NMR spectrum consists of two singlets at δ 1.2 and δ 3.6. Which is the most likely structure of the compound?arrow_forward(a) When cis-1-bromo-2-methylcyclohexane undergoes an E2 reaction, two products (cycloalkenes) are formed. What are these two cycloalkenes, and which would you expect to be the major product? Write conformational structures showing how each is formed. (b) When rans-1-bromo-2-methylcyclohexane reacts in an E2 reaction, only one cyclo- alkene is formed. What is this product? Write conformational structures showing why it is the only product.arrow_forward
- Compounds A and B are isomers having the molecular formula C4H8O3. Identify A and B on the basis of their 1H NMR spectra.Compound A: δ 1.3 (3H, triplet); 3.6 (2H, quartet); 4.1 (2H, singlet); 11.1 (1H, broad singlet)Compound B: δ 2.6 (2H, triplet); 3.4 (3H, singlet); 3.7 (2H triplet); 11.3 (1H, broad singlet)arrow_forwardCompounds Y and Z are isomers with the molecular formula C10H12O. The IR spectrum of each compound shows a strong absorption band near 1710 cm−1 . The 1H NMR spectra of Y and Z are given below. Propose structures for Y and Z.arrow_forwardThe reaction of 3,4-dimethyl-3-hexanol (3,4-dimethylhexan-3-ol) with HBr generates compound A as the major product. Treatment of compound A with a strong base gives two isomers of compound B as the major product, along with one isomer of compound C and one isomer of compound D as minor products, all of which have one double-bond equivalent. Identify compounds A, B, C, and D and give their names. By what mechanism does the reaction of 3,4-dimethyl-3-hexanol with HBr occur? By what mechanism does the reaction of A with strong base to form B occur? Propose reaction conditions for an alternative, one-step method for converting 3,4-dimethyl-3-hexanol directly to compound B.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning