
Concept explainers
(a)
Interpretation: The mechanism for the given chemical transformation is to be interpreted.
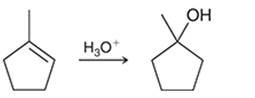
Concept introduction:
(b)
Interpretation: The mechanism for the given chemical transformation is to be interpreted.
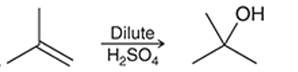
Concept introduction:
Alkenes are unsaturated hydrocarbons with at least one double bond between the carbon atoms. The acid-catalyzed hydration reactions of alkene involve the reaction of an alkene with
(c)
Interpretation: The mechanism for the given chemical transformation is to be interpreted.
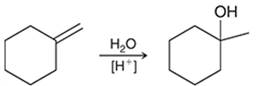
Concept introduction:
Alkenes are unsaturated hydrocarbons with at least one double bond between the carbon atoms. The acid-catalyzed hydration reactions of alkene involve the reaction of an alkene with
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
ORGANIC CHEMISTRY (LL) W/ACCESS
- 10arrow_forwardIdentify the reagents necessary to accomplish each of the following transformations.arrow_forwardTreatment of W with CH3Li, followed by CH3I, affords compound Y (C7H14O) as the major product. Y shows a strong absorption in its IR spectrum at 1713 cm−1, and its 1H NMR spectrum is given below. (a) Propose a structure for Y. (b) Draw a stepwise mechanism for the conversion of W to Y.arrow_forward
- Identify reagents that can be used to achieve each of the following transformationsarrow_forwardDevise a concise synthesis for the following transformations. Clearly show the reagent and productfor each step. Each synthesis can be accomplished in two stepsarrow_forwardPlease complete parts A and Barrow_forward
- Draw a stepwise mechanism for the following reaction, one step in the synthesis of the cholesterol-lowering drug pitavastatin, marketed in Japan as a calcium salt under the name Livalo.arrow_forwardDraw a stepwise mechanism for the following reaction, which results in the synthesis of bisphenol F (R = H), an additive used in a variety of packaging materials. Bisphenol F is related to BPA (bisphenol A, R = CH3), a reagent used to harden some plastics, now removed from certain baby products because of its estrogen-like activity that can disrupt endocrine pathways.arrow_forwardDraw the mechanism for the following transformationarrow_forward
- How would you prepare tamoxifen, a drug used in the treatment of breast cancer, from the following ketone and any other reagents needed?arrow_forwardDraw a stepwise mechanism for the following reaction, one step in the synthesis of the cholesterol-lowering drug pitavastatin, marketed in Japan as a calcium salt under the name Livalo.arrow_forwardDraw a stepwise, detailed mechanism for the following reaction: 2 .CH3 ملی LOH H2SO4 + 2 CH3OH HOarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





