
Concept explainers
The compound butyric acid gives rancid butter its characteristic odor. (7.1, 7.2)
a. If black spheres are carbon atoms, white spheres are hydrogen atoms, and red spheres are oxygen atoms, what is the molecular formula of butyric acid?
b. What is the molar mass of butyric acid?
c. How many grams of butyric acid contain 3.28 x 1O atoms of oxygen?
d. How many grams of carbon are in 5.28 g of butyric acid?
e. Butyric acid has a density of 0.959 g/mL at 20 °C. How many moles of butyric acid are contained in 1.56 mL of butyric acid?
f. Identify the bonds C—-C, C- H, and C— 0 in a molecule of butyric acid as polar covalent or nonpolar covalent.
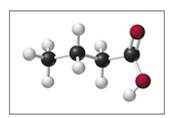
Butyrlc acid produces the characteristic odor of rancid butter.
Want to see the full answer?
Check out a sample textbook solution
Chapter 9 Solutions
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
- The active ingredient of Benadryl© Chesty Forte Cough Liquid is Guaiphenesin an expectorant drug used to assist the expectoration ('bringing up') of phlegm from the airways in acute respiratory tract infections. A 200.0 mL bottle of Benadryl© Chesty Forte Cough contains 4.00 g of Guaiphenesin (C10H14O4). If the recommended dose for an adult is 350 mg of Guaiphenesin. How many moles of Guaiphenesin does this equal? (answer to 2 significant figures)arrow_forwardComplete combustion of 7.30 g of a hydrocarbon produced 22.4 g of CO2 and 10.7 g of H2O. Divide the number of moles of carbon and hydrogen by the smallest of the two values. Report these ratios to three significant figures. (Having most trouble with the sig figs.)arrow_forwardDraw a potential energy diagram for a system in which the forward reaction has Eact = +42 kcal/mol and the reverse reaction has Eact = +28 kcal/mol. a. Is the forward process endothermic or exothermic? ["", ""] b. What is the value for ΔH for this reaction? Please insert an image of your workarrow_forward
- Find ΔHrxn for the following reaction given the table of data below: 3 H2(g) + O3(g) → 3 H2O(g) Reaction ΔHrxn 2 H2(g) + O2(g) → 2 H2O(g) -483.6 kJ/mol 3 O2(g) → 2 O3(g) +285.4 kJ/molarrow_forwardInitial Temp of HCl: 22.96 C Final Temp of HCl: 28.30 C Moles of HCl: .05 moles Moles of NaOH: .05 moles Cs water: 4.184 J/g C Accepted delta Hrxn: -55.8 kJ/mol 1. what is the qwater. pls include formula. 2. calculate delta Hrxnn using the moles, temp and cswater 3. calculate delta Hrxn % errorarrow_forwarda cereal contains 11 grams of sucrose per 60g of cereal. How many grams of cereal must be eated to consume 0.0424 moles of sucrosearrow_forward
- Dichloromethane (CH2Cl2)(CH2Cl2), used as a solvent in the decaffeination of coffee beans, is prepared by reaction of methane (CH4)(CH4) with chlorine. How many grams of dichloromethane result from reaction of 2.51 kgkg of methane if the yield is 44.5 %% ?CH4Methane(g)+2Cl2Chlorine(g)→CH2Cl2Dichloromethane(l)+2HCl(g)arrow_forwardif ammonia is added with cupric oxide,how many moles of gas is produced from 6 moles of cupric oxidearrow_forward3. Syngas is a mixture of carbon monoxide, carbon dioxide, and water vapor that is used for making fertilizer, methanol, and other products. It is produced by heating fossil fuels under low-oxygen conditions to promote incomplete combustion. Write an equation for the complete combustion of the gasoline component cyclopentane, C5H10 (l) and the incomplete combustion of cyclopentane with 20% of the carbon appearing as CO(g).arrow_forward
- Na2SO4 • XH2O If I have 0.548 g of the hydrate after weighing and 0.272 g of H2O was lost after heating the hydrate, what is the value of x for the hydrate?arrow_forwardWhat type of reaction and how much energy is involved in a reaction of boron trichloride with hydrochloric acid? B2H6(g) +6Cl2(g) ⟷2BCl3(g) + 6HCl(g) ΔH= -755.4 kJ/mole Group of answer choices Exothermic with ΔH = -755.4 kJ/mole. Exothermic with ΔH = +755.4 kJ/mole. Endothermic with ΔH = -755.4 kJ/mole. Endothermic with ΔH = +0.001324 kJ/mole. Endothermic with ΔH = +755.4 kJ/mole.arrow_forwardA student reacted 3.01g salicylic acid with 5.98mL acetic anhydride and isolated 2.85g Asprin. A) Calculate moles of salicylic acid used. B) Calculate the mass of acetic anhydride used. C) Calculate moles of acetic anhydride used. D) using moles of limiting reactant present, calculate the theoretical yield of aspirin. E) Calculate the percent yield of aspirin synthesis.arrow_forward
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
