
Organic Chemistry, 12e Study Guide/Student Solutions Manual
12th Edition
ISBN: 9781119077329
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9, Problem 18PP
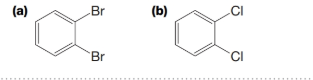
PRACTICE PROBLEM 9.18
What are the expected ratios of the
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
10.10 Show graphically how you would expect AH
reaction [M(H,O)6]²+ + 6 NH3
[M(NH3)6]²+ + 6 H,O to vary for the first transition
for
the
series (M = Sc through Zn).
We have a pink crystals of an ionic compound of an f- block element (M). The crystals house the octahydrate [ M(H2O)8]3+ and an anion which is not chloride and does not luminesce under UV. (A) First, draw and label a plausible solid-state geometry for the hydrated cation. (B)Provide ALL potential identities for the f- block element, M, based on the information supplied above, defending your choices and discussing the applicable chemical concepts. (C)What are the potential solid state MCI3 geometries and coordination numbers for ALL of the components proposed in section (B)? Explain briefly why, focusing on the key chemical principles. (D) In ion - exchange chromatography, which of the trivalent f-block ions indicated in component (B) would you anticipate to elute first? Explain your response, emphasising the important chemical concepts.
Write the equations involved in the following reactions:(i) Wolff-Kishner reduction(ii) Etard reaction
Chapter 9 Solutions
Organic Chemistry, 12e Study Guide/Student Solutions Manual
Ch. 9 - Prob. 1PPCh. 9 - PRACTICE PROBLEM 9.2 What compound with molecular...Ch. 9 - PRACTICE PROBLEM 9.3
Using the method of Section...Ch. 9 - PRACTICE PROBLEM 9.4 How many signals would each...Ch. 9 - Prob. 5PPCh. 9 - Prob. 6PPCh. 9 - PRACTICE PROBLEM 9.7
The relative chemical shifts...Ch. 9 - Prob. 8PPCh. 9 - PRACTICE PROBLEM 9.9 Propose a structure for...Ch. 9 - PRACTICE PROBLEM 9.10
What is the dihedral angle...
Ch. 9 - PRACTICE PROBLEM 9.11 Draw the most stable chair...Ch. 9 - Prob. 12PPCh. 9 - PRACTICE PROBLEM 9.13 How many signals would you...Ch. 9 - Prob. 14PPCh. 9 - Prob. 15PPCh. 9 - Prob. 16PPCh. 9 - Prob. 17PPCh. 9 - PRACTICE PROBLEM 9.18
What are the expected ratios...Ch. 9 - Prob. 19PPCh. 9 - How many 1H NMR signals (not peaks) would you...Ch. 9 - How many 13C NMR signals would you predict for...Ch. 9 - Prob. 22PCh. 9 - Prob. 23PCh. 9 - Prob. 24PCh. 9 - Compound Q has the molecular formula C7H8. The...Ch. 9 - 9.26 Explain in detail how you would distinguish...Ch. 9 - Compound S (C8H16) reacts with one mole of bromine...Ch. 9 - A compound with molecular formula C4H8O has a...Ch. 9 - In the mass spectrum of 2, 6-dimethyl-4-heptanol...Ch. 9 - Prob. 30PCh. 9 - What are the masses and structures of the ions...Ch. 9 - Prob. 32PCh. 9 - Ethyl bromide and methoxybenzene (shown below)...Ch. 9 - 9.34 The homologous series of primary amines, ,...Ch. 9 - Propose a structure that is consistent with each...Ch. 9 - 9.36 Propose structures for compounds E and F....Ch. 9 - 9.37 Use the NMR and IR data below to propose a...Ch. 9 - 9.38 When dissolved in , a compound (K) with the...Ch. 9 - Compound T (C5H8O) has a strong IR absorption band...Ch. 9 - Prob. 40PCh. 9 - Deduce the structure of the compound that gives...Ch. 9 - Deduce the structure of the compound that gives...Ch. 9 - The 1H NMR spectrum of a solution of 1,...Ch. 9 - Acetic acid has a mass spectrum showing a...Ch. 9 - The 1H NMR peak for the hydroxyl proton of...Ch. 9 - The 1H NMR study of DMF (N, N-dimethylformamide)...Ch. 9 - 9.48 The mass spectra of many benzene derivatives...Ch. 9 - Prob. 49PCh. 9 - 1. Given the following information, elucidate the...Ch. 9 - Two compounds with the molecular formula C5H10O...
Additional Science Textbook Solutions
Find more solutions based on key concepts
For the 3pz and 4dxz hydrogen-like atomic orbitals, sketch the following: a. The radial function R b. The radia...
Inorganic Chemistry
If a compound has a molecular ion with an odd-numbered mass, then the compound contains an odd number of nitrog...
Organic Chemistry
The glycine cleavage system is a group of four enzymes that together catalyze the following reaction: glycine+T...
Organic Chemistry (8th Edition)
16.43 The following pictures represent solutions at various stages in thetitration of a weak diprotic acid with...
Chemistry (7th Edition)
The NF bond is more polar than the NH bond: but NF3 has a smaller dipole moment than NH3. Explain this curious ...
Organic Chemistry (9th Edition)
Strengths of Brnsted-Lowry Acids and Bases Consider the following: CO/ is a weaker base than hydroxide ion, and...
Chemistry: The Molecular Nature of Matter
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Describe the general trends in the following properties of first series 3d of transition elements: (i) number of oxidation states exhibited (ii) formation of oxometal ions.arrow_forwardWhat is the possible unknown compound based on the IR Spectrum, please give detailed explanation on why it is the compound and also give detailed explanation on each of the other compounds unknown compound melting point is 170 0Carrow_forward5. (a) The mass spectrum of rhodium carbonyl complex includes peaks at 748 amu, 720 amu and decreasing every 28 amu down to 412 amu and then at 309 amu, 206 amu and 103 amu. Identify, the complex (Rh = 103). %3Darrow_forward
- (a) (b) The quantum yield of fluorescence for anthracene is seen to be higher than that of biphenyl. Draw the two compounds and explain why this is so. The UV spectrum of isoquinoline has four 2 max values at 318, 305, 266 and 218 nm. Draw the compound and suggest what you would expect to happen if acid was added to solution and the spectrum is retaken.arrow_forward(18) shown below? Which of the following options represents the HOMO for the pentadienyl anion, 80000 (A) (B) Sa (C) (D)arrow_forwardProvide the correct IUPAC name for Sr₃P₂. (I) (II) (III) (IV) hexa- hepta- tri- tetra- octa- di- penta- mono- phosphite strontium phosphorus potassium phosphonium phosphide sulfur phosphate hydrate acid Deletearrow_forward
- Explain the following observations :(a) Silver atom has completely filled d-orbitals (4d10) in its ground state, yet it is regarded as a transition element.(b) E0 value for Mn3+ /Mn2+ couple is much more positive than Cr3+/Cr2+.arrow_forward(d) Exchange of H₂O for D₂O in [Cr(H₂O)]³+ is extremely slow (k = 2.6 x 10-6 s-¹ at 25°C), while for [Cr(H₂O)]²+ it is very fast (k = 1.2 x 108 s1 at 25°C). Account for these observations and suggest the most likely electronic configuration of [Cr(H₂O)6]²+.arrow_forwardIf you needed to know the oxidation states of the elements on the surfaće of a material, which of the following techniques would yield the most complete information? Defend your answer. (a) AFM (b) STM (c) SEM (d) XRF spectroscopy (e) X-ray photoelectron spectroscopy (f) Auger electron spectroscopy You are choosing the electron gun for a TEM instrument. Your goal is to get the best resolution possible. Which type of electron gun should you purchase?arrow_forward
- a) Illustrate the transfer of electrons from the ligand to metal ion in hexaaquairon(III) ion, (Fe(H,O).J°*. b) Distinguish between an absorption spectrum and a calibration curve in terms of its y- axis and x-axis. c) Differentiate between the cause of real and chemical deviations.arrow_forwardThe frequency of the symmetrical M-O stretching vibration of the octahedral aqua ions [M(OH2)6]2+ increases along the series Ca2+<Mn2+<Ni2+. How does this trend relate to acidity?arrow_forward7. Write defect equations for the following reactions: (a) MgO doped with Li₂O (b) Slight reduction of PrO₂ (c) TiO₂ doped with Ga₂03 (d) LaCoO3 doped with CaO (e) MgAl2O4 doped with V₂O3 to form Mg(Al2-xVx)204arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY