
Concept explainers
Answers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book.
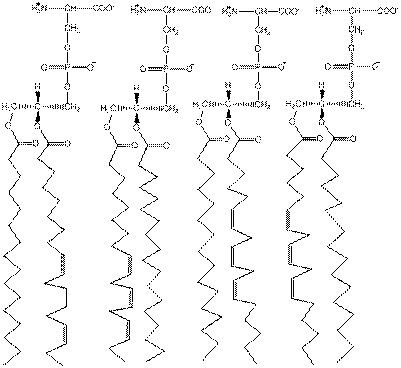
Understanding the Occurrence of Natural Phospholipids In problem 1 (b) ill Chapter 8, you weft asked to draw all the possible phosphate-dylserme isomers that form be formed from palmitic and linolenie acids. Which of the PS isomers aft not likely ll> be found in biological membranes?
Interpretation:
The PS isomer which is not likely to be found in biological membranes should be determined.
Concept Introduction:
There are two wide kinds of isomers: stereoisomers and constitutional.
Constitutional isomers vary in connectivity and bonding, whereas stereoisomers vary in 3-D orientation.
Answer to Problem 1P
Two structures having linolenic acid is connected to the initial carbon and palmitic acid is attached to the second carbon.
Explanation of Solution
Lipid is in proximity within biological membranes. Formation of double bond results in the bend in the chain which creates space among lipids.
Phosphatidyl serine molecules contain the big group on glycerol’s third carbon. Due to this group, the 2nd and 3rd carbons of glycerol contain more space as compared to the first carbon.
The fatty acid that attaches to the first carbon is usually a saturated fatty acid. An unsaturated fatty acid attaches to the second carbon.
Phosphatidylserines contain the phosphate bond in the serine residue on the 3rd carbon of glycerol.
Linoleic acid contains two common forms, thus, there are four kinds of different phosphatidylserines created with linoleic and palmitic acid.
The structure is shown as follows:
Want to see more full solutions like this?
Chapter 9 Solutions
EBK BIOCHEMISTRY
- Answers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Solving the Sequence of an Oligopeptide From Sequence Analysis Data Amino acid analysis of ail oligopeptide seven residues long gave The following fads were observed: a. Trypsin treatment had no apparent effect. b. The phenylthiohydantoin released by Lid mini degradation was c. Brief chymotrypsin treatment yielded several products, including a dipeptide and a tetrapeptide. The amino acid composition of the tetrapeptide was Leu, Lyi. and Met. d. Cyanogen bromide treatment yielded a dipeptide, a tetrapeptide, and free Lys. What is the amino acid sequence of this heptapeptide?arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Draw the Titration Curve for a Weak Acid and Determine its pKa from the Titration Curve When a 0.1 M solution of a weak acid was titrated with base, the following results were obtained: Plot the results of this titration and determine the pK a of the weak acid from your graph.arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Calculating the Molecular Weight and Subunit Organization of a Protein From Its Metal Content The element molybdenum {atomic weight 95.95) constitutes 0.0K if the weight of nitrate reductase. If the molecular weight of nitrate reductase is 240,000, what is its likely quaternary structure?arrow_forward
- Answers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Determining the Branch Points and Reducing Ends of Amylopectin A 0.2-g sample of amylopectin was analyzed to determine the fraction of the total glucose residues, that are branch points in the structure. The sample was exhaustively methylated and then digested, yielding 50-mol of 2,3-dimethylgluetose and 0.4 mol of 1,2,3,6- letramethylglucose. What fraction of the total residues are branch points? I low many reducing ends does this sample of amylopectin have?arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Interpreting Kinetics Experiments from Graphical Patterns The following graphical patterns obtained from kinetic experiments have several possible interpretations depending on the nature of the experiment and the variables being plotted. Give at least two possibilities for each.arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Assessing the pH Dependence of Poly-L-Glutamate Structure Poly-L glutamate adopts an tr-helical structure at low pH but becomes a random coil above pH 5. Explain this behavior.arrow_forward
- Answers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. (Research Problem) The Nature and Roles of Linear Motifs in Proteins In addition to domains and modules, there are other significant sequence patterns in proteins—known as linear motifs—that are associated with a particular function. Consult the biochemical literature to answer the following questions: 1. What are linear motifs? 2. How are they different from domains?. 3. What are their functions? 4. How can they be characterized? 5. There are several papers that are good starting points for this problem. Neduva, V., and Russell, R., 2005. Linear motifs: evolutionary interaction switches. FEBS Letters 579:3342-3345. Gibson, T., 2009. Cell regulation: determined to signal discrete cooperation. Trends in Biochemical Sciences 34:471-482. Diella, K. Haslam, N., Chica., C. et aL, 2009. Understanding eukaryotic linear motifs and their role in cell signaling and regulation. Frontiers of Bioscience 13:6580-6603.arrow_forwardAnswers to all problems are at the end of this book.. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Calculating the Composition of Anomeric Sugar Mixtures -D-Glucose has a specific notation, []220, of + 112.20. whereas -D-glucose has a specific notation of +18.70. What is the composition of a mixture of - and -D-glucose, which has a specific notation of 83 .U0?arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. The Strength of Weak Forces Determines the Environmental Sensitivity of Living Cells Why does the central role of weak forces in biomolecular interactions restrict living systems to a narrow range of environ¬mental conditions? (Section 1.4)arrow_forward
- Answers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. A Rule of Thumb for Amino Acid Content in Proteins The simple average molecular weight of the 20 common amino adds is 138, but most biochemists use 110 when estimating the number of amino acids in a protein of known molecular weight. Why do you Suppose this is? (Hint: There are two contributing factors to the answer. One of them will be apparent from a brief consideration of the amino acid compositions of common proteins. See, for example, Figure 5.16 of this text.)arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Understanding the Phase Transitions of Membrane Phospholipids Discuss the effects on the lipid phase transition of pure dimyristoyl phosphatidylcholine vesicles of added (a) divalent cations, (b) cholesterol, (c) distearoyl phosphatidylscrine. (d) dioleoyl phosphatidylcholine, and (e) integral meinbrane proteins.arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Phosphorylation of Proteins Introduces New Properties Phosphoproteins are formed when a phosphate group is esterified to an —OH group df a Ser, Thr, or Tyr side chain. At typical cellular pH values, this phosphate group bears two negative charges (— OPO32:_). Compare this side-chain modification to the 2U side chains of the Common amino acids found in proteins and comment on the novel properties that it introduces into side-chain possibilities.arrow_forward
 BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
