
Concept explainers
Deduce the structure of the compound that gives the following
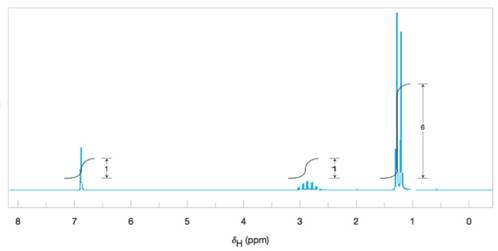
Figure 9.46 The
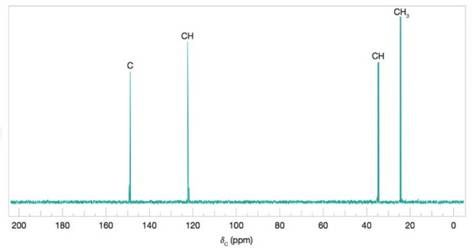
Figure 9.47 A simulated broadband proton-decoupled
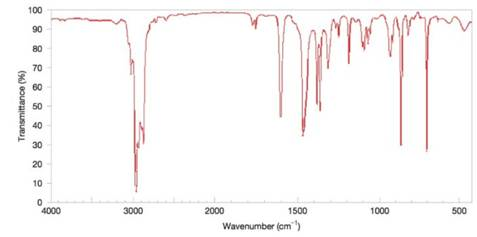
Figure 9.48 The IR spectrum for Problem 9.42. (SDBS, National Institute of Advanced Industrial Science and Technology)
Want to see the full answer?
Check out a sample textbook solution
Chapter 9 Solutions
ORGANIC CHEMISTRY-ETEXT REG ACCESS
Additional Science Textbook Solutions
Chemistry: The Central Science (13th Edition)
Organic Chemistry As a Second Language: Second Semester Topics
Chemistry: Structure and Properties (2nd Edition)
Basic Chemistry (5th Edition)
- Propose structures for compounds that fit the following descriptions: (a) A hydrocarbon with seven lines in its 13C NMR spectrum (b) A six-carbon compound with only five lines in its 13C NMR spectrum (c) A four-carbon compound with three lines in its 13C NMR spectrumarrow_forwardThe 1H-NMR spectrum of ethanol shows a triplet at 1.23 ppm, a singlet at 2.61 pm, and a quartet at 3.69 ppm. Assign each signal to the protons it corresponds to in the molecule. Explain the splitting pattern observed for each signal.arrow_forwardPart C Look at the NMR spectrum and list the structural characteristics you can determine from that spectrum. 200 180 160 140 120 100 80 60 40 20 13C NMR (CH, (CH, (CH) |(CH) CDCI (CH) (C) SOH SOH 7.18 708 6.18 6.08 CHCI, 10 8 (ppm) Part D Look at the set of spectra as a group and propose a structure the compound.arrow_forward
- (3) These natural compounds will have different numbers of 1H-NMR signals in different NMR solvents like CDC13 and methanol-d4. How many signals do you anticipate for each compound respectively in CDC13 and methanol-d4? LOCH 3 OHC. vanillin OH LOH acetoaminophen HO thymolarrow_forward1. How many proton signals would you expect to see in the ¹H-NMR spectrum of the following molecule? For each of the proton signals, predict the splitting pattern, assuming that you can see only 3-bond splitting.arrow_forwardPlease show work 1. Compound A has molecular formula C5H10O. It shows three signals in the 1H-NMR spectrum - a doublet of integral 6 at 1.1 ppm, a singlet of integral 3 at 2.14 ppm, and a quintet of integral 1 at 2.58 ppm. Suggest a structure for A and explain your reasoning.arrow_forward
- Below are the ¹H NMR spectrum of triphenylmethanol, benzophenone, and bromobenzene. Identify the compound corresponding to each ¹H NMR spectrum and draw the structure next to the ¹H NMR spectrum. Assign ALL peaks in each of the three ¹H NMR spectra. Hint: Conjugated systems (benzophenone) including an electronegative atom will cause a more downfield shift of ring protons in ¹H NMR compared with non-conjugated systems (bromobenzene). 8 8 8 7 7 7 6 6 6 5 5 5 4 PPM 4 PPM 4 PPM 3 3 3 2 2 2 1 1 1 0 0 0arrow_forwardHow many peaks would appear in the ¹3C NMR spectrum of this compound?arrow_forwardKindly provide the structure for each spectra given. I am expecting three structures in total.arrow_forward
- The 1H-NMR spectrum of 1,3-propanediol (HO-CH2-CH2-CH2-OH) shows a quintet at 1.81 ppm, a singlet at 2.75 pm, and a triplet at 3.83 ppm. Assign each signal to the protons it corresponds to in the molecule. Explain the splitting pattern observed for each signal.arrow_forwardThe 13C NMR spectra (decoupled 13C 1H spectrum and 3 DEPT spectra) and 1H NMR spectra of the compound in CDC13are shown below. Its molecular formula is C6H1002. Explain why the answer to this spectrum is this molecule -3495.6 1D Carbon DEPT45 ամուր DEPT90 DEPT135 www 200 7.0 1.0 18866 Loave 6.5 -3474.9 3473.2 7.0 180 -3459.3 6.0 elllle ppm 1.0 160 5.5 - 2922.2 2920.5 02162 5.9 wanando 5.0 140 ма 4.5 2906.6 2904.9 2903.2 2901.5 2.0 B 120 4.0 100 f1 (ppm) 3.5 3.0 2091.3 2084.2 2077.1 2069.9 CDCI 3 80 2.5 2.0 60 1.5 6879 933.6 3.0 926.7 924.9 5.8 ppm 4.2 ppm Figure 2: ¹H NMR spectra recorded in CDC13. 3.0 1.9 40 ppm ppm سال I 20 L 0 *** 1.3 ppmarrow_forwardThe mass spectrum of compound C, with a molecular formula C8H7BrO2 is shown below. 1.Draw the most likely ion fragment for the signals at m/z 155, 159, 183, and 185.Explain why these signals appear as pairs with almost similar intensities in the mass spectrum. 2.Identify and draw the structure of compound C.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


