
Concept explainers
The chapter sections lo review are shown in parentheses at the end of each problem
If red spheres represent oxygen atoms, blue spheres represent nitrogen atoms, and all the molecules are gases, (9.2, 9.4)
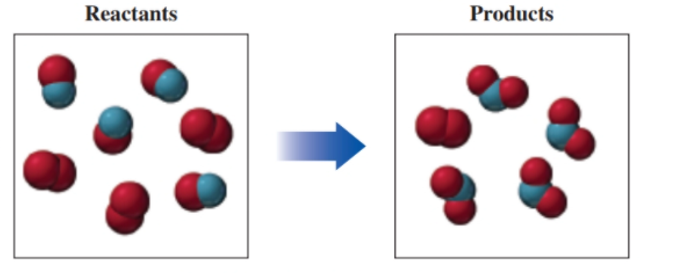
a. write a balanced equation for the reaction.
b. identify the limiting reactant.
(a)
Interpretation : The balanced chemical equation for the given reaction should be determined.
Concept Introduction : The balance equation for any chemical reaction represents the molecules of reactant and products with same number of atoms at both sides of the equation. The reactant and product molecules must be shown with simplest whole number coefficients.
Answer to Problem 47UTC
Explanation of Solution
Here, in reactant box there are 4 molecules of
(b)
Interpretation : The limiting reactant should be determined.
Concept Introduction : Limiting reactant is the reactant for a chemical reaction which is present in limited amount. Since it is present in limited amount therefore it determines the amount of product formed during the chemical reaction.
Answer to Problem 47UTC
NO is limiting reactant for the given reaction.
Explanation of Solution
Here, in reactant box there are 4 molecules of
Want to see more full solutions like this?
Chapter 9 Solutions
Basic Chemistry
- Ammonia will react with fluorine in the gaseous state to produce dinitrogen tetrafluoride and hydrogen fluoride according to the equation: α NH3(g) + β F2(g) → γ N2F4(g) + δ HF(g) The best values for α, β, γ and δ are: A. 2, 1, 1, 6 B. 2, 3, 1, 6 C. 2, 5, 1, 6 D. 2, 10, 1, 6 E. 2, 6, 1, 6arrow_forwardUsing the information in the table below, calculate the heat of released from the following reaction in kJ per g of NO. ( Mw. Of NO = 30.01 g/mol). 4NH3(g) + 5O2 (g) 6H2O(g) + 4 NO(g) NH3 H2O NO Hfº (kJ/mol) - 46.1 -241.8 + 90.3arrow_forwardA 27 g aluminum foil pan is used to roast vegetables. The pan is put into a cold oven at 22 oC . How much energy in cal is absorbed by the pan after cooking at 232 oC (450. oF) for 25 minutes?arrow_forward
- Draw a potential energy diagram for a system in which the forward reaction has Eact = +42 kcal/mol and the reverse reaction has Eact = +28 kcal/mol. a. Is the forward process endothermic or exothermic? ["", ""] b. What is the value for ΔH for this reaction? Please insert an image of your workarrow_forwardWhat type of reaction and how much energy is involved in a reaction of boron trichloride with hydrochloric acid? B2H6(g) +6Cl2(g) ⟷2BCl3(g) + 6HCl(g) ΔH= -755.4 kJ/mole Group of answer choices Exothermic with ΔH = -755.4 kJ/mole. Exothermic with ΔH = +755.4 kJ/mole. Endothermic with ΔH = -755.4 kJ/mole. Endothermic with ΔH = +0.001324 kJ/mole. Endothermic with ΔH = +755.4 kJ/mole.arrow_forwardWhen the reaction shown below is performed using: A + B --> C + D The temperature changes from 25.0 degrees Celsius at the start to 19.0 degrees Celsius at the end of the reaction. a. Is the reaction endothermic or exothermic? b. What is the value of q for the reaction if the mass of the reaction was 54.4 grams ?arrow_forward
- A student reacted 1.01g salicylic acid with 2.06mL acetic anhydride and isolated 1.40g aspirin. A) Calculate moles of acetic anhydride used. B) Using the moles of limiting reactant present, calculate the theoretical yield of aspirin. C) Calculate the percent yield of the aspirin synthesis.arrow_forwardA jeepney plying the route of Blumentrit - Pier consumes 8 liters of diesel per 100 kilometers. How much carbon dioxide does the jeepney liberate?arrow_forwardDuring the combustion of 5.00 gg of octane, C8H18C8H18, 239.5 kcalkcal (1002 kJkJ) is released. what is the balanced equation for the combustion reaction? How much energy (in kJkJ) is released by the combustion of 1.21 molmol of C8H18C8H18 ? How many moles of octane must be burned to release 442.4 kcalkcal ? How many kilocalories are released by the combustion of 18.3 gg of C8H18C8H18 ?arrow_forward
- A marshmallow is burned in a soda can calorimeter, and heats up 0.255 kg of water by 12.9°C. The calorimeter was calibrated and had a calorimetry constant of 8.50 kcal/kg・°C. Given that carbohydrates contain 4.00 kcal/g, calculate the mass in g of the sugar contained in the marshmallow. Assume that the marshmallow contains no fat or protein.arrow_forwardWhen 2 moles of HBr(g) react with Cl2(g) to form HCl(g) and Br2(g) according to the following equation, 81.1 kJ of energy are evolved.2HBr(g) + Cl2(g)=2HCl(g) + Br2(g) What is the value of q?arrow_forwardCalculate the heat required when 2.50 mol of A reacts with excess B and A2B according to the reaction:2A + B + A2B → 2AB + A2Given(on the photo):arrow_forward
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

