
Concept explainers
Predict the bond angles for the following molecules:
Interpretation:
The bond angles of the given molecules are to be determined.
Concept introduction:
Electron domain is the number of bonds and lone pair of electrons around the central atom.
According to VSEPR, the electron domains repel each other and arrange themselves as far apart as possible in space.
Molecular geometry represents the arrangement of bonded atoms. If no lone pairs are present, then the molecular geometry is the same as the electron domain geometry.
The repulsion in lone pair–lone pair is greater than that in lone pair–bond pair, which in turn is greater than that in bond pair–bond pair. The repulsion due to a double bond is more than that due to single bonds.
Answer to Problem 75AP
Solution:
a)
b)
c)
d)
e)
f) Less than
g) Less than
h)
Explanation of Solution
a)
The Lewis structure of
is as follows:
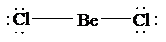
In this structure, the
atom is bonded to two chlorine atoms by single bonds, and there is no lone pair on
atom. According to VSEPR, two electron domains are arranged on opposite sides to give a linear shape, with a bond angle of
b)
The Lewis structure of
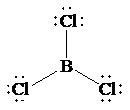
In this structure, the
atom is bonded to three chlorine atoms by single bonds, and there is no lone pair on
atom. According to VSEPR, three electron domains are arranged in a trigonal planar shape with a bond angle of
c)
The Lewis structure of
is as follows:
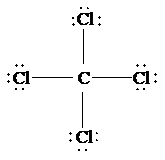
In this structure, the
atom is bonded to four chlorine atoms by single bonds, and there is no lone pair on
atom. According to VSEPR, four electron domains are arranged in a tetrahedral shape with a bond angle of
d)
The Lewis structure of
is as follows:
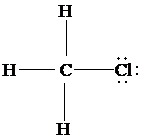
In this structure, the
atom is bonded to three hydrogen atoms and one chlorine atom by single bonds, and there is no lone pair on
atom. According to VSEPR, four electron domains are arranged in a tetrahedral shape with a bond angle of
e)
The Lewis structure of
is as follows:
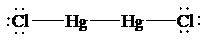
In this structure, each
atom is bonded to one chorine atom, and to each other. Thus, there are two electron domains around each
atom. Thus, the shape is linear with a bond angle of
f)
The Lewis structure of
is as follows:
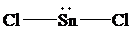
In this structure, the
atom is bonded to two chlorine atoms by single bonds, and there is a lone pair on
atom. According to VSEPR, three electron domains are arranged in a trigonal planar shape with a bond angle of
atom, the shape of
is bent with a bond angle less than
g)
The Lewis structure of
is as follows:
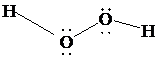
In this structure, each
atom is bonded to one hydrogen atom, and to each other. There are two lone pairs on each oxygen atom. Thus, there are four electron domains around each oxygen atom. According to VSEPR, four electron domains have a tetrahedral shape. Due to the presence of two lone pairs, the shape is bent with a bond angle less than
h)
The Lewis structure is as follows:
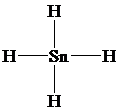
In this structure, the
atom is bonded to four chlorine atoms by single bonds, and there is no lone pair on
atom. According to VSEPR, four electron domains are arranged in a tetrahedral shape with a bond angle of
Want to see more full solutions like this?
Chapter 9 Solutions
ALEKS 360 CHEM ACCESS CARD
- In each of the following molecules, a central atom is surrounded by a total of three atoms or unshared electron pairs: SnCl2, BCl3, SO2. In which of these molecules would you expect the bond angle to be less than 120? Explain your reasoning.arrow_forwardHistidine is an essential amino acid that the body uses to form proteins. The Lewis structure of histidine follows. What are the approximate values for bond angles 1 through 5 (indicated on the structure by blue numbers)?arrow_forwardIt is possible to write a simple Lewis structure for the SO42- ion, involving only single bonds, which follows the octet rule. However, Linus Pauling and others have suggested an alternative structure, involving double bonds, in which the sulfur atom is surrounded by six electron pairs. (a) Draw the two Lewis structures. (b) What geometries are predicted for the two structures? (c) What is the hybridization of sulfur in each case? (d) What are the formal charges of the atoms in the two structures?arrow_forward
- Could the anion Li2 exist? What is the ions bond order?arrow_forwardIndicate which molecules are polar and which are nonpolar. (a) SeO2 (b) N2O (N is the central atom) (c) SCl4arrow_forwardWhat are the bond angles predicted by the VSEPR model about the carbon atom in the formate ion, HCO2? Considering that the bonds to this atom are not identical, would you expect the experimental values to agree precisely with the VSEPR values? How might they differ?arrow_forward
- The cations O2+ and N2+ are formed when molecules of O2 and N2 are subjected to intense, high-energy solar radiation in Earths upper atmosphere. Write the electron configuration for O2+. Predict its bond order and magnetic behavior.arrow_forwardFor the anions (Na2S2O4), (LiBH4), (Ca (PF6)2), ((NH4)2PO3F) and (Na2S2O3) propose the most probable Lewis structure, its molecular geometry, bond angles, formal charges, and the hybridization of each central atom.arrow_forwardDraw the Lewis structure for acetamide (CH3CONH2), an organic compound, and determine the geometry about each interior atom. Experiments show that the geometry about the nitrogen atom in acetamide is nearly planar. Which resonance structure can account for the planar geometry about the nitrogen atom?arrow_forward
- The molecular structure of the molecule sulfur cyanide trifluoride, CF3NS, is characterized by the following bond lengths: C-N/ 116 picometers, S-C/ 174 picometers, and S-F/ 160 picometers. Using the observed bond lengths as a guide, draw a realistic lewis structure for sulfur cyanide tirfluoride and assign formal charges to each atom. Are there any resonance structures? Predict the hybridization of the C , N, and S atoms.arrow_forwardElectrons in bonds are negatively charged and therefore repel each other. If you had a molecule of formula AB 2 , with A being the central atom, what would you predict the 3D shape to be to allow maximum distance between the two B atoms bonded to A?arrow_forwardBy looking at the Lewis Structure alone, how do I know whether CH2Cl2 is polar? I know its electron domain geometry is tetrahedral and I thought so would its molecular geometry. Therefore I assumed it should be non-polar, what am I missing?arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





