
Concept explainers
Interpretation:
H2 reacts with O2 and produced water vapors. The molecular picture of product and the left-over reactant needs to be determined.
Concept Introduction:
The reaction in which the reactant is totally consumed when the reaction is completed is
known as limiting reactant.
Answer to Problem 7STP
The left-over reactant is O2 molecules.
Explanation of Solution
The given reaction is shown below:
The balanced
H2 molecule represented as follows:

O2 molecules represented as follows:

The molecular picture is shown below:
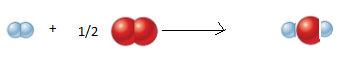
As
According to the balanced reaction; 1 molecule of H2 reacts with ½ molecule of O2 and produces 1 molecule of H2O. In the given picture there are 6 molecules of H2 are present and 6 molecules of O2 are present. Therefore; 6 molecules of H2 reacts with 3 molecules of O2 but there are 6 molecules of O2 present. So, H2 act as a limiting reactant. Hence, 6 molecules of H2 will react with 3 molecules of O2 and will produce 6 molecule of water and the remaining 3 molecules of oxygen will be present as they don’t react.
Therefore; the left-over reactant is O2.
The molecular picture is shown below:
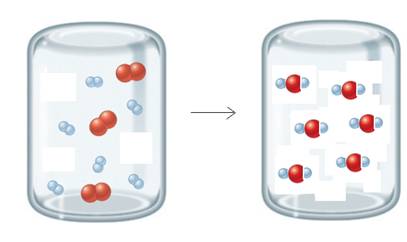
The left-over reactant is O2 molecules
Chapter 9 Solutions
World of Chemistry
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





