
Concept explainers
Interpretation:
The mixture of helium (red) and argon (blue) when the temperature is lowered to
Concept Introduction:
Ideal gas equation is an equation that is describing the state of an imaginary ideal gas.
Where,
P is the pressure of the gas
V is the volume
n is the number of moles of gas
R is the universal gas constant
T is the temperature
Charles law,
Volume of the container is directly proportional to Temperature of the container
Where,
V=Volume
T=Temperature
Answer to Problem 9.26CP
The volume of the container will decreases when the temperature decreases.
Explanation of Solution
Given,
According to Charles law,
Volume of the container is directly proportional to Temperature of the container
Where,
V=Volume
T=Temperature
So the volume of the container will reduce to half.
When T=
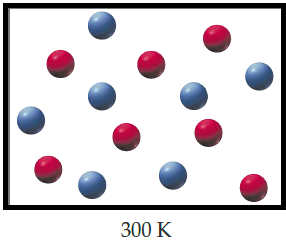
Figure 1
When T=
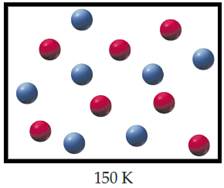
Figure 2
The mixture of helium (red) and argon (blue) when the temperature is lowered to
Want to see more full solutions like this?
Chapter 9 Solutions
General Chemistry: Atoms First, Books a la Carte Edition; Modified Mastering Chemistry with Pearson eText -- ValuePack Access Card -- for General Chemistry: Atoms First (2nd Edition)
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





