
(a)
To determine: The structural formula for
Interpretation: The structural formula for
Concept introduction: Structural formulas are used to describe the arrangement of atoms, groups or substituents in a molecule, whereas molecular formula describes the total number and the type of atoms present in a molecule. The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because IUPAC names are applied at international level and it comprises suffix, prefix, numbers and other priority rules.
(a)
Answer to Problem 9.26SP
The structural formula for
Explanation of Solution
The structural formula for
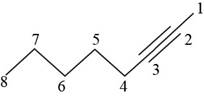
Figure 1
The structural formula for
(b)
To determine: The structural formula for
Interpretation: The structural formula for
Concept introduction: Structural formulas are used to describe the arrangement of atoms, groups or substituents in a molecule, whereas molecular formula describes the total number and the type of atoms present in a molecule. The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because IUPAC names are applied at international level and it comprises suffix, prefix, numbers and other priority rules.
(b)
Answer to Problem 9.26SP
The structural formula for
Explanation of Solution
The structural formula for
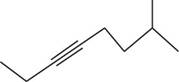
Figure 2
The structural formula for
(c)
To determine: The structural formula for
Interpretation: The structural formula for
Concept introduction: Structural formulas are used to describe the arrangement of atoms, groups or substituents in a molecule, whereas molecular formula describes the total number and the type of atoms present in a molecule. The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because IUPAC names are applied at international level and it comprises suffix, prefix, numbers and other priority rules.
(c)
Answer to Problem 9.26SP
The structural formula for
Explanation of Solution
The structural formula for
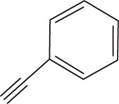
Figure 3
The structural formula for
(d)
To determine: The structural formula for
Interpretation: The structural formula for
Concept introduction: Structural formulas are used to describe the arrangement of atoms, groups or substituents in a molecule, whereas molecular formula describes the total number and the type of atoms present in a molecule. The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because IUPAC names are applied at international level and it comprises suffix, prefix, numbers and other priority rules.
(d)
Answer to Problem 9.26SP
The structural formula for
Explanation of Solution
The structural formula for
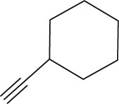
Figure 4
The structural formula for
(e)
To determine: The structural formula for
Interpretation: The structural formula for
Concept introduction: Structural formulas are used to describe the arrangement of atoms, groups or substituents in a molecule, whereas molecular formula describes the total number and the type of atoms present in a molecule. The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because IUPAC names are applied at international level and it comprises suffix, prefix, numbers and other priority rules.
(e)
Answer to Problem 9.26SP
The structural formula for
Explanation of Solution
The structural formula for
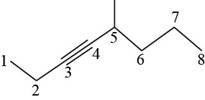
Figure 5
It is conferred form the above structure that the structural formula for
(f)
To determine: The structural formula for
Interpretation: The structural formula for
Concept introduction: Structural formulas are used to describe the arrangement of atoms, groups or substituents in a molecule, whereas molecular formula describes the total number and the type of atoms present in a molecule. The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because IUPAC names are applied at international level and it comprises suffix, prefix, numbers and other priority rules.
(f)
Answer to Problem 9.26SP
The structural formula for
Explanation of Solution
The structural formula for
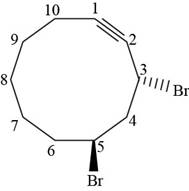
Figure 6
The stereochemistry along with structural formula for
(g)
To determine: The structural formula for
Interpretation: The structural formula for
Concept introduction: Structural formulas are used to describe the arrangement of atoms, groups or substituents in a molecule, whereas molecular formula describes the total number and the type of atoms present in a molecule. The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because IUPAC names are applied at international level and it comprises suffix, prefix, numbers and other priority rules.
(g)
Answer to Problem 9.26SP
The structural formula for
Explanation of Solution
The structural formula for
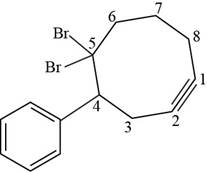
Figure 7
The structural formula for
(h)
To determine: The structural formula for
Interpretation: The structural formula for
Concept introduction: Structural formulas are used to describe the arrangement of atoms, groups or substituents in a molecule, whereas molecular formula describes the total number and the type of atoms present in a molecule. The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because IUPAC names are applied at international level and it comprises suffix, prefix, numbers and other priority rules.
(h)
Answer to Problem 9.26SP
The structural formula for
Explanation of Solution
The structural formula for
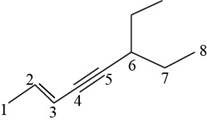
Figure 8
The structural formula for
(i)
To determine: The structural formula for
Interpretation: The structural formula for
Concept introduction: Structural formulas are used to describe the arrangement of atoms, groups or substituents in a molecule, whereas molecular formula describes the total number and the type of atoms present in a molecule. The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because IUPAC names are applied at international level and it comprises suffix, prefix, numbers and other priority rules.
(i)
Answer to Problem 9.26SP
The structural formula for
Explanation of Solution
The structural formula for
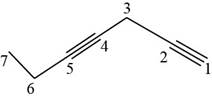
Figure 9
The structural formula for
(j)
To determine: The structural formula for
Interpretation: The structural formula for
Concept introduction: Structural formulas are used to describe the arrangement of atoms, groups or substituents in a molecule, whereas molecular formula describes the total number and the type of atoms present in a molecule. The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because IUPAC names are applied at international level and it comprises suffix, prefix, numbers and other priority rules.
(j)
Answer to Problem 9.26SP
The structural formula for
Explanation of Solution
The structural formula for
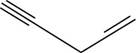
Figure 10
The structural formula for
(k)
To determine: The structural formula for
Interpretation: The structural formula for
Concept introduction: Structural formulas are used to describe the arrangement of atoms, groups or substituents in a molecule, whereas molecular formula describes the total number and the type of atoms present in a molecule. The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because IUPAC names are applied at international level and it comprises suffix, prefix, numbers and other priority rules.
(k)
Answer to Problem 9.26SP
The structural formula for
Explanation of Solution
The structural formula for
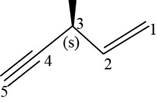
Figure 11
The structural formula for
Want to see more full solutions like this?
Chapter 9 Solutions
ORGANIC CHEMISTRY
- 4. MAINIDEA Compare and contrast alkyl halides and aryl halides. 5. Draw structures for the following molecules. a. 2-chlorobutane c. 1,1,1-trichloroethane b. 1,3-difluorohexane d. 1-bromo-4-chlorobenzene 6. Define functional group and name the group present in each of the following structures. Name the type of organic compound each substance represents. a. CH3CH,CH2OH b. CH;CH,F c. CH;CH,NH2 7. Evaluate How would you expect the boiling points of propane and 1-chloropropane to compare? Explain your answer. d. CH3C- OH 8. Interpret Scientific Illustrations Examine the pair of substituted hydrocarbons illustrated at right, and decide whether it represents a pair of optical isomers. Explain your answer.arrow_forwardShow how to convert 1- Butene into these compounds. a. Butane b. 2- Butanol c. 2- Bromobutane d. 1,2- Dibromobutanearrow_forward6. Write the structure of the following compounds. a. 4 - ethyl - 2 - hexoneb. 2-ethyl - 3 – pentanoic acidarrow_forward
- Name one structural isomer created by changing theposition of one or more halogen atoms in each alkylhalide.a. 2-chloropentane c. 1,3-dibromocyclopentaneb. 1,1-difluropropane d. 1-bromo-2-chloroethanearrow_forward1. 1-Ethoxy-2-methylpropane A. Acid anhydride B. Acid chloride C. Ether D. None of the choices 2. C8H16 A. Alkane B. Alkene C. Cycloalkane D. Two of the choices 3. How many CH2 is present in this compound? (Please refer to the timage attached.) A. 6 B. 5 C. 4 D. 3arrow_forwardBased on the image attached, what is the name of the compound? A. 3-chloro-2-ethylpyrimidine B. 3-chloro-2-ethyl-1,4-diazacyclohexane C. 3-chloro-2-ethylcyclohexan-1,4-diamine D. 2-chloro-3-ethyl-1,4-diazacyclohexanearrow_forward
- Explanation Questions 1. Why do compounds form different shapes? ( 2. What does the specific heat capacity of a substance measure? 3. Explaining how removing reactant will affect the equilibrium of a reaction. ( 4. What does a standard reduction table represent, and how is it used? ( 5. What is the current model of electrons in an atom? Problem Solving Questions 1. Draw the following compounds:/ a. butan-2-ol b. 4-methyl pentan-2-onearrow_forwardWhich is not a structural isomer of hexyne. a. 2-hexyne b. 3-hexyne c. 2,2-dimethylpentane d. 4-methyl-1-pentyne e. 2,3 - dimethylbutane.arrow_forwardA hydrocarbon containing one or more -C=C- bonds. a. addition reaction b. aliphatic compound c. alkene d. alkyne e. aromatic hydrocarbon f. hydration g. hydrogenation h. monomer i. phenyl group j. polycyclic aromatic hydrocarbon k. polymerarrow_forward
- What is the product ? A. 3-methylhexane B. cis-sec-butene C. cis-3-methyl-2-hexene D. trans-ethyl-2-pentenearrow_forwardDraw skeletal structures for the following: a. 5-ethyl-2-methyloctane b. 1,3-dimethylcyclohexane c. 2,3,3,4-tetramethylheptane d. propylcyclopentane e. 2-methyl-4-(1-methylethyl)octane f. 2,6-dimethyl-4-(2-methylpropyl)decanearrow_forward6. Any organic compound that contains a benzene ring or similar feature. O a. addition reaction Ob. aliphatic compound Oc. alkene d. alkyne Oe. aromatic hydrocarbon Of. hydration g. hydrogenation h. monomer Oi. phenyl group j. polycyclic aromatic hydrocarbon k. polymer I. unsaturated hydrocarbonarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





