
Modified Masteringchemistry With Pearson Etext -- Standalone Access Card -- For Basic Chemistry Format: Access Card Package
5th Edition
ISBN: 9780134270357
Author: Timberlake, Karen C.
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9, Problem 9.52UTC
If green spheres represent chlorine atoms and red spheres represent oxygen atoms, and all the molecules are gases. (9.2, 9.4, 9.5)
Reactants Actual products
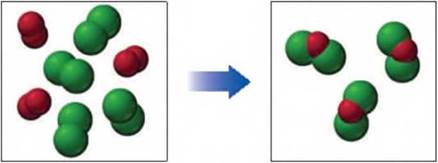
- write a balanced equation for the reaction
- identify the limiting reactant
- from the diagram of the actual products that result, calculate the percent yield for the reaction
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Liquid menthonal (CH4O) which is used as a cooking fuel, burns with oxygen gas to produce the gases carbon dioxide and water. The reaction produces 363 kJ of heat per mole of methonol.
a) write a balanced equation for the reaction includig the heat of the reaction
b) is the reaction endothermic or exothermic?
c) How many moles of O2 must react with 0.450 mole of CH4O?
d) How many grams of CO2 are produced when 78.0 g of Ch4O reacts?
A 27 g aluminum foil pan is used to roast vegetables. The pan is put into a cold oven at 22 oC . How much energy in cal is absorbed by the pan after cooking at 232 oC (450. oF) for 25 minutes?
a 20 mg sample CxHy is burned in oxygen to produce 60 mg of carbon dioxide and 32 mg of water. calculate the value of x and y.
Chapter 9 Solutions
Modified Masteringchemistry With Pearson Etext -- Standalone Access Card -- For Basic Chemistry Format: Access Card Package
Ch. 9.1 - Calculate the total mass of the reactants and the...Ch. 9.1 - Calculate the total mass of the reactants and the...Ch. 9.2 - 9.3 Write all of the mole-mole factors for each of...Ch. 9.2 - Prob. 9.4QAPCh. 9.2 - Prob. 9.5QAPCh. 9.2 - Prob. 9.6QAPCh. 9.2 - Prob. 9.7QAPCh. 9.2 - Ammonia is produced by the reaction of nitrogen...Ch. 9.2 - Prob. 9.9QAPCh. 9.2 - Prob. 9.10QAP
Ch. 9.3 - Sodium reacts with oxygen to produce sodium oxide....Ch. 9.3 - Nitrogen gas reacts with hydrogen gas to produce...Ch. 9.3 - Prob. 9.13QAPCh. 9.3 - lron (III) oxide reacts with carbon to give iron...Ch. 9.3 - Nitrogen dioxide and water react to produce nitric...Ch. 9.3 - Calcium cyanamide, CaCN2 , reacts with water to...Ch. 9.3 - When solid lead(II) sulfide reacts with oxygen...Ch. 9.3 - When the gases dihydrogen sulfide and oxygen...Ch. 9.4 - Prob. 9.19QAPCh. 9.4 - A clock maker has 15 clock faces. Each clock...Ch. 9.4 - Prob. 9.21QAPCh. 9.4 - Prob. 9.22QAPCh. 9.4 - 9.23 For each of the following reactions, 20.0 g...Ch. 9.4 - For each of the following reactions. 20.0 g of...Ch. 9.4 - Prob. 9.25QAPCh. 9.4 - Prob. 9.26QAPCh. 9.5 - 9.27 Carbon disulfide is produced by the reaction...Ch. 9.5 - Prob. 9.28QAPCh. 9.5 - Prob. 9.29QAPCh. 9.5 - Prob. 9.30QAPCh. 9.5 - Prob. 9.31QAPCh. 9.5 - Prob. 9.32QAPCh. 9.6 - In an exothermic reaction, is the energy of the...Ch. 9.6 - Prob. 9.34QAPCh. 9.6 - Prob. 9.35QAPCh. 9.6 - Classify each of the following as exothermic or...Ch. 9.6 - Prob. 9.37QAPCh. 9.6 - Prob. 9.38QAPCh. 9.6 - Prob. 9.39QAPCh. 9.6 - Prob. 9.40QAPCh. 9.6 - Prob. 9.41QAPCh. 9.6 - Calculate the enthalpy change for the reaction...Ch. 9.6 - Prob. 9.43QAPCh. 9.6 - Prob. 9.44QAPCh. 9 - Prob. 9.45FUCh. 9 - Prob. 9.46FUCh. 9 - Prob. 9.47UTCCh. 9 - Prob. 9.48UTCCh. 9 - Prob. 9.49UTCCh. 9 - Prob. 9.50UTCCh. 9 - Prob. 9.51UTCCh. 9 - If green spheres represent chlorine atoms and red...Ch. 9 - Prob. 9.53UTCCh. 9 - Prob. 9.54UTCCh. 9 - When ammonia NH3 reacts with fluorine F2 , the...Ch. 9 - Gasohol is a fuel that contains ethanol C2H6O that...Ch. 9 - When hydrogen peroxide H2O2 is used in rocket...Ch. 9 - Propane gas, C3H8 , reacts with oxygen to produce...Ch. 9 - When 12.8 g of Na and 10.2 g of Cl2 react, what is...Ch. 9 - If 35.8 g of CH4 and 75.5 g of S react, how many...Ch. 9 - Pentane gas, C5H12 , reacts with oxygen to produce...Ch. 9 - When nitrogen dioxide NO2 from car exhaust...Ch. 9 - The gaseous hydrocarbon acetylene, C2H2 , used in...Ch. 9 - The equation for the decomposition of potassium...Ch. 9 - When 28.0 g of acetylene reacts with hydrogen....Ch. 9 - When 50.0 g of iron(III) oxide reacts with carbon...Ch. 9 - Nitrogen and hydrogen combine to form ammonia....Ch. 9 - Prob. 9.68AQAPCh. 9 - The equation for the reaction of nitrogen and...Ch. 9 - The equation for the reaction of iron and oxygen...Ch. 9 - Each of the following is a reaction that occurs in...Ch. 9 - Each of the following is a reaction that occurs in...Ch. 9 - Prob. 9.73CQCh. 9 - Prob. 9.74CQCh. 9 - Prob. 9.75CQCh. 9 - Prob. 9.76CQCh. 9 - Prob. 9.77CQCh. 9 - Prob. 9.78CQCh. 9 - Prob. 9.79CQCh. 9 - Prob. 9.80CQCh. 9 - Prob. 9.81CQCh. 9 - Prob. 9.82CQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Nitrogen oxide (NO) has been found to be a key component in many biological processes. It also can react with oxygen to give the brown gas NO2. When one mole of NO reacts with oxygen, 57.0 kJ of heat is evolved.A. Write the thermochemical equation for the reaction between one mole of nitrogen oxide and oxygen.B. Is the reaction exothermic or endothermic?C. What is H when 5.00 grams of nitrogen oxide reacts?D. How many grams of nitrogen oxide must react with an excess of oxygen to liberate ten kilojoules of heat?arrow_forwardΔHr for N2(g) + 3H2(g) <------> 2NH3 (g) is -92.2 kJ/mol reaction a) What will be the enthalpy change if 0.200 mol of N2 reacts? b) How many moles of H2 must be consumed if 0.200 moles of N2 reacts? c) How many moles of NH3 can be formed if 0.200 moles of N2 reacts? d) Calculate ΔHr for the reaction in questions b) and c). Pays careful attention to units .arrow_forwardWhat is the mass of SO3 when -8753 kJ of energy are used according to the following equation?2S + 3O2 → 2SO3 ∆H° = -791.4kJ Is this Endothermic or Exothermicarrow_forward
- an important flavor component of vanilla extract is vanillin C8H8O3 molar mass = 152.15 g/mol when vanillin burns in a bomb calorimeter with a heat capacity of 12.16 kJ/ celsius the temp increases from 24.56 celsius to 30.60 celsius. if the heat of combustion of vanillin is -3760 kJ/mol what mass of vanillin (in g) was combusted? answer to two decimal placesarrow_forwardWhen 1.0 g of gasoline burns, it releases 11 kcal. The density of gasoline is 0.74 g/mL. (3.4, 3.6) How many megajoules are released when 1.0 gal of gasoline burns? If a television requires 150 kJ/h to run, how many hours can the television run on the energy provided by 1.0 gal of gasoline?arrow_forwardThe reaction between nitrogen and oxygen gases require the absorption of 43 kJ of energy per mole of nitrogen according to the following chemical equation, N2(g) + O2(g) -> 2 NO(g). How many grams of NO are produced in the reaction if 328 kJ of heat are absorbed by the reaction?arrow_forward
- Using the information in the table below, calculate the heat of released from the following reaction in kJ per g of NO. ( Mw. Of NO = 30.01 g/mol). 4NH3(g) + 5O2 (g) 6H2O(g) + 4 NO(g) NH3 H2O NO Hfº (kJ/mol) - 46.1 -241.8 + 90.3arrow_forwardAn Iron ore sample contains Fe2O3 together with other substances. Reaction of the ore with CO produces iron metal. __Fe2O3(s)+__CO(g)-->__Fe(s)+__CO2(g) a) balance this equation b) Calculate the grams of CO that can react with .350kg of Fe2O3 c) Calculate the grams of Fe and the grams of CO2 formed when .350kg of Fe2O3 reactsarrow_forward21 Consider the combustion of in oxygen gas to produce carbon dioxide gas and water vapor. In an experiment, 0.1063 g of C₅H₈ is combusted to produce enough heat to raise the temperature of 150.0 g of water by 7.632 °C. C₅H₈ (l) + 7 O₂ (g) → 5 CO₂ (g) + 4 H₂O (g) e If -4790 J of heat was produced from the combustion of 0.561 moles of C5H8, what is the enthalpy change (in kJ/mol) for the combustion of C5H8?arrow_forward
- an important flavor component of vanilla extract is vanillin C8H8O3 molar mass = 152.15 g/mol when vanillin burns in a bomb calorimeter with a heat capacity of 8.60 kJ/°C the temp increases from 25.97°C to 31.18°C. if the heat of combustion of vanillin is -3760 kJ/mol what mass of vanillin (in g) was combusted? answer to two decimal placesarrow_forwardwhen a 0.55-g of olive oil is burned in a calorimeter the hear released increases the temperature of 360 g of water from 22.7 c to 36.4 c . what is the energy value for the olive oil in kcal/g?arrow_forwardMethane, CH4, reacts with oxygen to produce carbon dioxide, water, and heat. CH4(g) + 2O2(g) . CO2(g) + 2H2O(l) DH = -890.3 kJ What is the value of DH if 5.00 g of CH4 is combusted? a. -157 kJ b. -277 kJ c. -445 kJ d. -714 kJ e. -1.43 ÅL 104 kJarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning
Calorimetry Concept, Examples and Thermochemistry | How to Pass Chemistry; Author: Melissa Maribel;https://www.youtube.com/watch?v=nSh29lUGj00;License: Standard YouTube License, CC-BY