
Concept explainers
Interpretation:
Whether the connectivity of
Concept Introduction:
Covalent bond is defined as a bond is formed from mutual sharing of electrons between atoms. Lewis structures are representations of the covalent bond. In this, Lewis symbols show how the valence electrons are present in the molecule.
The steps to draw the Lewis structure of the molecule are as follows:
Step 1: Find the central atom and place the other atoms around it. The atom in a compound that has the lowest group number or lowest electronegativity considered as the central atom.
Step 2: Estimate the total number of valence electrons.
Step 3: Connect the other atoms around the central atoms to the central atom with a single bond and lower the value of valence electrons by 2 of every single bond.
Step 4: Allocate the remaining electrons in pairs so that each atom can get 8 electrons.
The formula to calculate formal charge of the atom is as follows:
The different structures can be drawn for the same molecule. Structures that minimize the amount of formal charge found on each atom are more stable than structures that place large amounts of formal charge on atoms.
The structures that have adjacent atoms with formal charges of the same sign are less stable. Lewis structures that show the smallest formal charges are stable. The structure that has negative formal charges on the more electronegative atoms are favored.
Explanation of Solution
For structure
The given compound is made up of carbon, hydrogen, and nitrogen atoms.
The rules applied to obtain the Lewis structure of
1. Write the skeleton structure.
There are three hydrogen atom, one carbon atom and a nitrogen atom. Therefore, 4 bonds are formed.
2. Calculate the total number of valence electrons.
The valence electron of nitrogen is calculated as follows:
The valence electron of carbon is calculated as follows:
The valence electron of hydrogen is calculated as follows:
The total number of valence electrons is calculated as follows:
3. Calculate the remaining electrons that are not used in skeleton structure.
The skeleton structure has 4 bonds. Therefore 8 electrons are used in bonds.
The remaining electrons are calculated as follows:
4 To obey the octet rule, carbon atom needs 2 electrons and nitrogen atom needs 4 electrons.
5. Satisfy the octet rule.
There are 4 remaining electrons. Multiple bonds can be formed. In this compound, an additional bond is needed to complete the structure. Also, remaining electrons are placed as lone pairs on atoms to satisfy octet.
The Lewis structure of
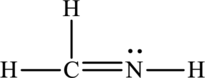
6. The Lewis structure is finished except for formal charges.
7. The formal charge on an atom in this Lewis structure can be calculated from the equation written as follows:
The formal charge on nitrogen atom is calculated as follows:
Substitute 5 for number of valence electrons, 2 for number of lone pairs and 6 for number of shared electrons in equation (1).
The formal charge on first hydrogen atom is calculated as follows:
Substitute 1 for number of valence electrons, 0 for number of lone pairs and 2 for number of shared electrons in equation (1).
The formal charge on second hydrogen atom is calculated as follows:
Substitute 1 for number of valence electrons, 0 for number of lone pairs and 2 for number of shared electrons in equation (1).
The formal charge on third hydrogen atom is calculated as follows:
Substitute 1 for number of valence electrons, 0 for number of lone pairs and 2 for number of shared electrons in equation (1).
The formal charge on carbon atom is calculated as follows:
Substitute 4 for number of valence electrons, 0 for number of lone pairs and 8 for number of shared electrons in equation (1).
In this Lewis structure, nitrogen, hydrogen and oxygen atom has formal charge 0.
The Lewis structure made from
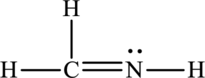
For structure
The given compound is made up of carbon, hydrogen, and nitrogen molecule.
The rules applied to obtain the Lewis structure of
1. Write the skeleton structure.
There are three hydrogen atom, one carbon atom, and nitrogen atom. Therefore, 4 bonds are formed.
2. Calculate the total number of valence electrons.
The valence electron of nitrogen is calculated as follows:
The valence electron of carbon is calculated as follows:
The valence electron of hydrogen is calculated as follows:
The total number of valence electrons is calculated as follows:
3. Calculate the remaining electrons that are not used in skeleton structure.
The skeleton structure has 4 bonds. Therefore 8electrons are used in bonds.
The remaining electrons are calculated as follows:
4 To obey the octet rule, carbon atom needs 2 electrons and nitrogen atom needs 4 electrons.
5. Satisfy the octet rule.
There are 4 remaining electrons. Multiple bonds can be formed. In this compound, an additional bond is needed to complete the structure. Also, remaining electrons are placed as lone pairs on atoms to satisfy octet.
The Lewis structure of
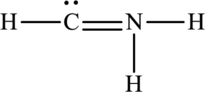
6. The Lewis structure is finished except for formal charges.
7. The formal charge on an atom in this Lewis structure can be calculated from the equation written as follows:
The formal charge on nitrogen atom is calculated as follows:
Substitute 5 for number of valence electrons, 2 for number of lone pairs and 6 for number of shared electrons in equation (1).
The formal charge on first hydrogen atom is calculated as follows:
Substitute 1 for number of valence electrons, 0 for number of lone pairs and 2 for number of shared electrons in equation (1).
The formal charge on second hydrogen atom is calculated as follows:
Substitute 1 for number of valence electrons, 0 for number of lone pairs and 2 for number of shared electrons in equation (1).
The formal charge on third hydrogen atom is calculated as follows:
Substitute 1 for number of valence electrons 0 for number of lone pairs and 2 for number of shared electrons in equation (1).
The formal charge on carbon atom is calculated as follows:
Substitute 4 for number of valence electrons, 0 for number of lone pairs and 8 for number of shared electrons in equation (1).
In this Lewis structure, nitrogen, hydrogen and oxygen atom has formal charge 0.
The Lewis structure made from
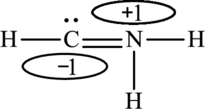
The
Hence,
Want to see more full solutions like this?
Chapter 9 Solutions
Chemistry
- Nitrosyl azide, N4O, is a pale yellow solid first synthesized in 1993. Write the Lewis structure for nitrosyl azide.arrow_forwardSuccessive substitution of F atoms for H atoms in the molecule NH3 produces the molecules NH2F, NHF2, and NF3. a. Draw Lewis structures for each of the four molecules. b. Using VSEPR theory, predict the geometry of each of the four molecules. c. Specify the polarity (polar or nonpolar) for each of the four molecules.arrow_forwardDraw Lewis structures for the following species. (The skeleton is indicated by the way the molecule is written.) (a) Cl2CO (b) H3C—CN (c) H2C—CH2arrow_forward
- Draw Lewis structures for the following species. (The skeleton is indicated by the way the molecule is written.) (a) Cl2CO (b) H3CCNarrow_forwardThe study of carbon-containing compounds and their properties is called organic chemistry. Besides carbon atoms, organic compounds also can contain hydrogen, oxygen, and nitrogen atoms (as well as other types of atoms). A common trait of simple organic compounds is to have Lewis structures where all atoms have a formal charge of zero. Consider the following incomplete Lewis structure for an organic compound called histidine (an amino acid), which is one of the building blocks of proteins found in our bodies: Draw a complete Lewis structure for histidine in which all atoms have a formal charge of zero.arrow_forwardA common trait of simple organic compounds is to have Lewis structures where all atoms have a formal charge of zero. Consider the following incomplete Lewis structure for an organic compound called methyl cyanoacrylate, the main ingredient in Super Glue. Draw a complete Lewis structure for methyl cyanoacrylate in which all atoms have a formal charge of zero.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





