
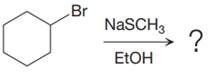
Concept explainers
(a)
Interpretation:
It is to be determined whether the given
Concept introduction:
Competing reactions can take place in kinetic or
Reactions that tend to take place under thermodynamic control are the ones in which a more stable but not necessarily the major product is formed.
Reversible reactions tend to take place under thermodynamic control, while irreversible reactions tend to take place under kinetic control.
Reactions like
The charge stability decides if the products are more stable than the reactants or vice versa. A reaction is irreversible if it’s
Answer to Problem 9.72P
The given reaction is irreversible.
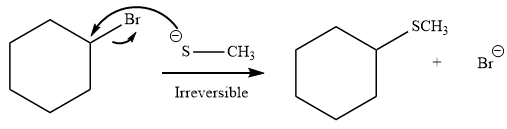
Explanation of Solution
The given reaction is:
It is mentioned that the reaction would follow the
The species
As the reaction is irreversible it would take place under kinetic control.
The charge stability decides if the products are more stable than the reactants or vice versa.
(b)
Interpretation:
It is to be determined whether the given
Concept introduction:
Competing reactions can take place in kinetic or thermodynamic control. Reactions that tend to take place under the kinetic control are the ones in which the major product is the one that forms the fastest.
Reactions that tend to take place under thermodynamic control are the ones in which a more stable but not necessarily the major product is formed.
Reversible reactions tend to take place under thermodynamic control, while irreversible reactions tend to take place under kinetic control.
Reactions like
The charge stability decides if the products are more stable than the reactants or vice versa. A reaction is irreversible if it’s
Answer to Problem 9.72P
The given reaction is reversible.
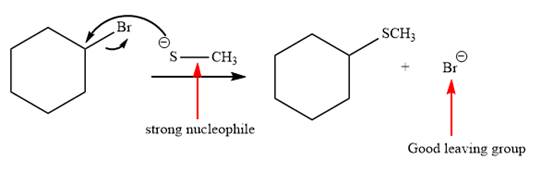
Explanation of Solution
The given reaction is:
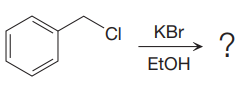
It is mentioned that the reaction would follow the
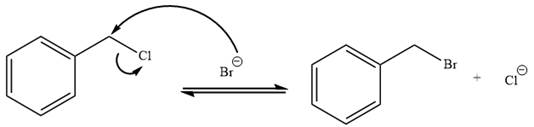
The charge stability decides if the products are more stable than the reactants or vice versa. The charged species on the left side is
This makes the reaction faster in the reverse direction than in the forward direction under standard conditions. Thus, this reaction is reversible.
The charge stability decides if the products are more stable than the reactants or vice versa.
(c)
Interpretation:
It is to be determined whether the given
Concept introduction:
Competing reactions can take place in kinetic or thermodynamic control. Reactions that tend to take place under the kinetic control are the ones in which the major product is the one that forms the fastest.
Reactions that tend to take place under thermodynamic control are the ones in which a more stable but not necessarily the major product is formed.
Reversible reactions tend to take place under thermodynamic control, while irreversible reactions tend to take place under kinetic control.
Reactions like
The charge stability decides if the products are more stable than the reactants or vice versa. A reaction is irreversible if it’s
Answer to Problem 9.72P
The given reaction is irreversible.
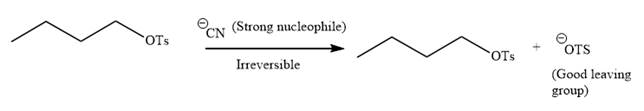
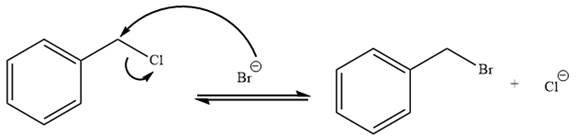
Explanation of Solution
The given reaction is:
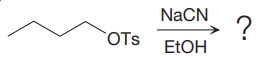
It is mentioned that the reaction would follow the
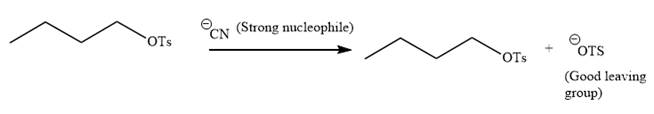
In the above reaction,
The charge stability decides if the products are more stable than the reactants or vice versa. The charged species on the left side is
Thus,
As the reaction is irreversible it would take place under kinetic control.
The charge stability decides if the products are more stable than the reactants or vice versa.
Want to see more full solutions like this?
Chapter 9 Solutions
ORGANIC CHEMISTRY PRINCIPLES & MECHANISM
- Explain how you can tell from the energy diagram that the reaction with the catalyst in Fig. 8.4 isfaster than the reaction without the catalyst.arrow_forwardDraw a detailed, step-wise mechanism for the reaction shown below. SHOW ALL BOND-FORMING AND BOND-BREAKING STEPS. SHOW ALL INTERMEDIATES. 1) NaOH 2) H3O* H3C H3C HOarrow_forwardAll I need help with, is everything about this process, please send help!arrow_forward
- What is the strongest nucleophile in this list? B OH D A B U Darrow_forwardConsider the two energy diagrams given below. B Reaction progress Reaction progress Which of the following is correct with respect to these diagrams? O A could represent an E2 reaction B could represent an E1 reaction A could represent an E1cb reaction O A could represent and E1 reaction B could represent an E1cb reaction Energyarrow_forwardThe reaction of methylpropene with HBr in ether gives one of the two products below as the major product. Br HBr Br ether Product A Product B Product would have a higher energy transition state for the formation of the intermediate leading to it. O A O B O Both products would have the same transition state.arrow_forward
- PLEASE HELParrow_forwardWhich is true of a free energy diagram for an SN1 eaction? It shows one transition state. It always shows that the reaction is net exothermic. It never shows intermediates. It always shows that the reaction is net endothermic. It always includes multiple energy barriers.arrow_forwardAnswer the question below the reaction. ta The reaction above proceeds through which type of mechanism? SN2 SN1 E1 E2 OH + Excess NH4C1 H₂SO4 + H₂Oarrow_forward
- ОН ОН BF3 Et,0 This reaction takes place via a pinacol rearrangement. Draw curved arrows to show the movement of electrons in this step of the reaction mechanism. Arrow-pushing Instructions :OHarrow_forward6. Predict the reactant (starting material) for the reaction and propose a mechanism. NaCN SN2 NaOH E2 CN a Ph Pharrow_forwardDraw the entire extended reaction mechanism (curly arrows) for this reaction но- OH HO HO OMe H OMe OMe HO HO OMearrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
