
CHEMISTRY:CENTRAL SCI.,V.1-PKG.>CUSTOM<
15th Edition
ISBN: 9781323444535
Author: Brown
Publisher: PEARSON C
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9, Problem 99AE
A typical amino acid with one amino group and one carboxylic acid group, such as serine, can exist in water in several ionic forms.
- Suggest the forms of the amino acid at low PH and at high pH.
- Amino acids generally have two pKa values, one in the range of 2 to 3 and the other in the range of 9 to 10. Serine, for example, has pKa values of 2.19 and 9.21. Using species such as acetic acid and ammonia as models, suggest the origin of the two pKa values.
- Glutamic acid is an amino acid that has three pKa’s: 2.10, 4.07, and 9.47. Draw the structure of glutamic acid, and assign each pKa to the appropriate part of the molecule.
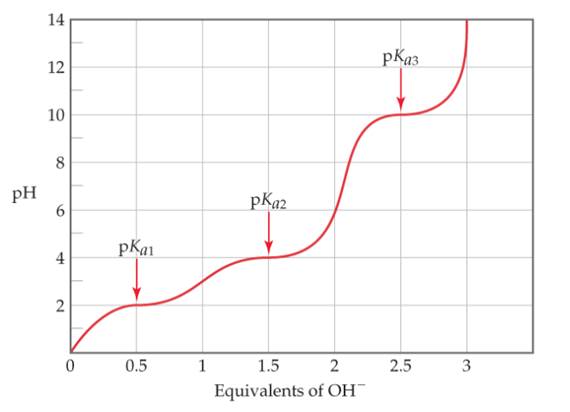
- An unknown amino acid is titrated with strong base, producing the following titration curve. Which amino acids are likely candidates for the unknown?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
A typical amino acid with one amino group and one carboxylic acid group, such as serine can exist in water in several iconic forms.
Suggest the forms of the amino acid at low pH and at a high pH.
Amino acids generally have two pKa values, one in the range of 2 to 3 and the other in the range of 9 to 10. Serene, for example, has pKa values of 2.91 and 9.21. Using species such as acetic acid and ammonia as models, suggest the origin of the two pKa values.
Glutamic acid is an amino acid that has three pKa’s: 2.10, 4.07, and 9.47. Draw the structure of glutamic acid and assign each pKa to the appropriate part of the molecule.
An unknown amino acid is titrated with strong base, producing the following titration curve. Which amino acid are likely candidates for the unknown?
. Describe the pH range of acceptable buffering behavior for the amino acids alanine, histidine, aspartic acid, and lysine.
A typical amino acid with one amino group and one carboxylicacid group, such as serine, can exist in water in several ionic forms. (a) Suggest the forms of the amino acidat low pH and at high pH. (b) Amino acids generally havetwo pKa values, one in the range of 2 to 3 and the other inthe range of 9 to 10. Serine, for example, has pKa values of2.19 and 9.21. Using species such as acetic acid and ammoniaas models, suggest the origin of the two pKa values.(c) Glutamic acid is an amino acid that has threepKa's: 2.10, 4.07, and 9.47. Draw the structure of glutamicacid, and assign each pKa to the appropriate part of themolecule. (d) An unknown amino acid is titrated withstrong base, producing the following titration curve.Which amino acids are likely candidates for the unknown?
Chapter 9 Solutions
CHEMISTRY:CENTRAL SCI.,V.1-PKG.>CUSTOM<
Ch. 9.2 - Prob. 9.1.1PECh. 9.2 - Prob. 9.1.2PECh. 9.2 - Prob. 9.2.1PECh. 9.2 - Prob. 9.2.2PECh. 9.2 - Prob. 9.3.1PECh. 9.2 - Prob. 9.3.2PECh. 9.3 - Prob. 9.4.1PECh. 9.3 - Prob. 9.4.2PECh. 9.5 - Prob. 9.5.1PECh. 9.5 - Prob. 9.5.2PE
Ch. 9.6 - Prob. 9.6.1PECh. 9.6 - Prob. 9.6.2PECh. 9.6 - Prob. 9.7.1PECh. 9.6 - Prob. 9.7.2PECh. 9.7 - Prob. 9.8.1PECh. 9.7 - Prob. 9.8.2PECh. 9.8 - Prob. 9.9.1PECh. 9.8 - Prob. 9.9.2PECh. 9 - Prob. 1DECh. 9 - Prob. 1ECh. 9 - Prob. 2ECh. 9 - Prob. 3ECh. 9 - Prob. 4ECh. 9 - Prob. 5ECh. 9 - Prob. 6ECh. 9 - Prob. 7ECh. 9 - Prob. 8ECh. 9 - Prob. 9ECh. 9 - Prob. 10ECh. 9 - Prob. 11ECh. 9 - Prob. 12ECh. 9 - Prob. 13ECh. 9 - Prob. 14ECh. 9 - Prob. 15ECh. 9 - Prob. 16ECh. 9 - Prob. 17ECh. 9 - Prob. 18ECh. 9 - Prob. 19ECh. 9 - Prob. 20ECh. 9 - Prob. 21ECh. 9 - Prob. 22ECh. 9 - Prob. 23ECh. 9 - Prob. 24ECh. 9 - Prob. 25ECh. 9 - Prob. 26ECh. 9 - Prob. 27ECh. 9 - Prob. 28ECh. 9 - Prob. 29ECh. 9 - Prob. 30ECh. 9 - Prob. 31ECh. 9 - Prob. 32ECh. 9 - Prob. 33ECh. 9 - Prob. 34ECh. 9 - Prob. 35ECh. 9 - Prob. 36ECh. 9 - Prob. 37ECh. 9 - Prob. 38ECh. 9 - Prob. 39ECh. 9 - Prob. 40ECh. 9 - Prob. 41ECh. 9 - Prob. 42ECh. 9 - Prob. 43ECh. 9 - Describe the intermediate that is thought to form...Ch. 9 - Prob. 45ECh. 9 - Prob. 46ECh. 9 - Prob. 47ECh. 9 - Prob. 48ECh. 9 - (a) Starting with the orbital diagram of a boron...Ch. 9 - Prob. 50ECh. 9 - Prob. 51ECh. 9 - Prob. 52ECh. 9 - Prob. 53ECh. 9 - Prob. 54ECh. 9 - Prob. 55ECh. 9 - Prob. 56ECh. 9 - Prob. 57ECh. 9 - Prob. 58ECh. 9 - Prob. 59ECh. 9 - Prob. 60ECh. 9 - Prob. 61ECh. 9 - Prob. 62ECh. 9 - Prob. 63ECh. 9 - Prob. 64ECh. 9 - In the formate ion, HCO2-, the carbon atom is the...Ch. 9 -
9.66 Consider the Lewis structure shown below....Ch. 9 - Prob. 67ECh. 9 - Prob. 68ECh. 9 - Prob. 69ECh. 9 - Prob. 70ECh. 9 - Prob. 71ECh. 9 - Prob. 72ECh. 9 - Prob. 73ECh. 9 - (a) What is the probability of finding an electron...Ch. 9 - Prob. 75ECh. 9 - Prob. 76ECh. 9 - Prob. 77ECh. 9 - Prob. 78ECh. 9 - Prob. 79ECh. 9 - Prob. 80ECh. 9 - Prob. 81ECh. 9 - Prob. 82ECh. 9 - Prob. 83ECh. 9 - Prob. 84ECh. 9 - Prob. 85AECh. 9 - Prob. 86AECh. 9 - Prob. 87AECh. 9 - Prob. 88AECh. 9 - Prob. 89AECh. 9 - Prob. 90AECh. 9 - Prob. 91AECh. 9 - Prob. 92AECh. 9 - Prob. 93AECh. 9 - Prob. 94AECh. 9 - Prob. 95AECh. 9 - Prob. 96AECh. 9 - Prob. 97AECh. 9 - Prob. 98AECh. 9 - A typical amino acid with one amino group and one...Ch. 9 - The azide ion, N3-, is linear with two N—N bonds...Ch. 9 - Prob. 101AECh. 9 - Prob. 102AECh. 9 - Prob. 103AECh. 9 - Prob. 104AECh. 9 - Prob. 105AECh. 9 - Prob. 106AECh. 9 - How many hydrogen atoms are in 2, 2-...Ch. 9 - Prob. 108AECh. 9 - Prob. 109AECh. 9 - Prob. 110AECh. 9 - Prob. 111AECh. 9 - Prob. 112AECh. 9 - Prob. 113IECh. 9 - Prob. 114IECh. 9 - Prob. 115IECh. 9 - Prob. 116IECh. 9 - Practice Exercise 2 Name the dipeptide and give...Ch. 9 - How many chiral carbon atoms are there in the...Ch. 9 - Prob. 119IECh. 9 - Prob. 120IECh. 9 - Prob. 121IECh. 9 - Prob. 122IE
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The pKa values of an amino acid are the following: pK1 = 2.18; pK2 = 8.95; pK3 = 10.53, what is the isoelectric point of the amino acid? Show the solution and express the your answer in three significant figures.arrow_forwardSketch a titration curve for the following amino acids and indicate the pKa values for all titratable groups. Also indicate the pH at which this amino acid has no net charge.arrow_forwardThere are many organic acids and bases in our cells, and their presence modifies the pH of the fluids inside them. It is useful to be able to assess the pH of solutions of acids and bases and to make inferences from measured values of the pH. A solution of equal concentrations of lactic acid and sodium lactate was found to have pH= 3.08. (a) What are the values of pKa and Ka of lactic acid? (b) What would the pH be if the acid had twice the concentration of the salt?arrow_forward
- How does the shape of a titration curve confirm the fact that the pH region of greatest buffering power for an amino acid solution is around its pK's? Name four factors (bonds or other forces) that contribute to stabilizing the native structure of a protein, and describe one condition or reagent that interferes with each type of stabilizing force. Why is silk fibroin so strong, but at the same time so soft and flexible?arrow_forwardConsider the amino acid and its pKa values:Lysine: pKa1= 2.18 pKa2= 8.95, pKaR=10.79 a. Calculate the isoelectric point.b. What will be the net charge on lysine at the isoelectric point?c. In what direction will the lysine move when placed in an electric field at a pH value of 6.68d. Which form of the lysine is the least soluble in water?e. Draw the structure of the lysine which is the least soluble in water?arrow_forwardThe first step in the procedure asks you to prepare five dilutions of the protein standard (BSA) in the range of 0.2 to 1.0 mg/ml. For each concentration, a volume of 1.0 mL should be prepared. Describe how you will prepare these standards using a stock solution of BSA that is 2.0 mg/ml.arrow_forward
- . For valine, methionine, GABA and proline, write the zwitterion. Using valine as an example, describe the behavior of the zwitterion in acidic (pH 2) and alkaline (pH 12) media.arrow_forwardGiven that C6H11COOH has a pKa = 4.8 and C6H11N + H3 has a pKa = 10.7, what pH would you make the water layer to cause both compounds to dissolve in it?arrow_forwardDraw the structures of the predominant forms of glutamic acid and tyrosine at pH 9.0 and pH 10.0. What is the second most abundant species at each pH?arrow_forward
- Explain why the pI of lysine is the average of the pKa values of its two protonated amino groups.arrow_forwardIf an amino acid has pKa1 = 2.71 and pKa2 = 9.31, at what pH value in aqueous solution would the predominant form of this amino acid be a zwitterion? Please explain your answer and show your work. a) 6.01 b) 2.71 c) 5.65 d) 12.02 e) 9.31arrow_forwardWhat is the actual chemical component for an anthocyanin (assume a cyanidin structure), and a carotenoid (assume a lutein structure), and indicate what is the common feature of these indicators that could act as an acid or base. (While the structures are complicated molecules, compare with acetic acid, CH3COOH, a weak acid).arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning
Biomolecules - Protein - Amino acids; Author: Tutorials Point (India) Ltd.;https://www.youtube.com/watch?v=ySNVPDHJ0ek;License: Standard YouTube License, CC-BY