
Loose Leaf for Chemistry in Context
9th Edition
ISBN: 9781260151763
Author: American Chemical Society
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9.11, Problem 9.33YT
Skill Building Meet DEHP
DEHP belongs to a common class of plasticizers called phthalates (THAL-ates). Phthalates are esters of phthal ic (THAL-ic) acid, an isomer of terephtha lic acid, one of the monomers used to synthesize PET.
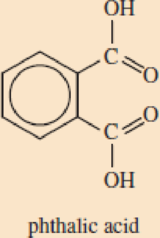
- a. Explain the meaning of the term ester.
- b. Below is the structural formula for DEHP. Circle the two ester groups in this molecule.
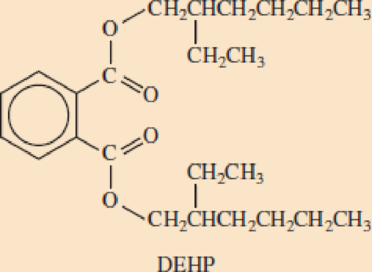
- c. Draw a structural formula for the alcohol that reacted with terephthalic acid to form this ester.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Skip Part
A
B
N. A
Functional group
alkyl halide
ketone
Check
B
Classification
3°
2°
N
O
HO
X
3
O Search
ions
Animations
Slide Show
Review
View
Help
A Viewing v
A
hello
Your turn – naming esters
Page 11 in b
H
H
H.
H C-C-0-C-C-H
H.
н н
Steps
1. Look at the alkyl group that came from the alcohol.
This is the first bit of the ester's name
2. Now look at the part that came from the carboxylic acid.
Swap its '-oic acid' ending for 'gate' to get the second bit of the na
3. Put the two parts together
Help Improve Office N
s.pptx - G.
hp
only
Search
ions
Animations
Slide Show
Review
View
Help
A Viewing
S Sha
A
A
而、川、
Page 5 in booklet
hello
Your turn
Name the following hydroxynitriles
H H
Но
CN
OH
Help Improve Office
A Notes e
1 - Aldehydes & K..
Microsoft - Google.
P L1 - Aldehydes & K.
hp
paus
bre
Scroll
f5
f6
17 C
f8
f9
f10
11
F12
(7)
&
18
8.
Chapter 9 Solutions
Loose Leaf for Chemistry in Context
Ch. 9.1 - Scientific Practices Tennis Anyone? Examine this...Ch. 9.3 - Prob. 9.2YTCh. 9.3 - Prob. 9.3YTCh. 9.4 - Prob. 9.4YTCh. 9.4 - Prob. 9.5YTCh. 9.4 - Prob. 9.6YTCh. 9.4 - Prob. 9.7YTCh. 9.4 - Prob. 9.8YTCh. 9.4 - Prob. 9.9YTCh. 9.5 - Prob. 9.10YT
Ch. 9.5 - Skill Building Benzene and Phenyl The difference...Ch. 9.5 - Prob. 9.13YTCh. 9.5 - Skill Building Polystyrene Possibilities Show the...Ch. 9.6 - Skill Building Esters and Polyesters You have seen...Ch. 9.6 - Prob. 9.16YTCh. 9.7 - Skill Building Kevlar Kevlar is a polyamide used...Ch. 9.8 - Prob. 9.20YTCh. 9.8 - Your Turn 9.22 Skill Building Burning a Plastic...Ch. 9.8 - Your Turn 9.23 Scientific Practices Landfill...Ch. 9.9 - Examine the values in Table 9.4 from the American...Ch. 9.9 - Prob. 9.25YTCh. 9.9 - Prob. 9.26YTCh. 9.9 - Prob. 9.28YTCh. 9.10 - Skill Building The Chemistry of PLA We dont show...Ch. 9.11 - Your Turn 9.31 Scientific Practices Glass or...Ch. 9.11 - Prob. 9.32YTCh. 9.11 - Skill Building Meet DEHP DEHP belongs to a common...Ch. 9 - Prob. 1QCh. 9 - Prob. 2QCh. 9 - Prob. 3QCh. 9 - Prob. 4QCh. 9 - Prob. 5QCh. 9 - Prob. 6QCh. 9 - Prob. 7QCh. 9 - Prob. 8QCh. 9 - Prob. 9QCh. 9 - Prob. 10QCh. 9 - Prob. 11QCh. 9 - Prob. 12QCh. 9 - Prob. 13QCh. 9 - Prob. 14QCh. 9 - Prob. 15QCh. 9 - Prob. 16QCh. 9 - Prob. 17QCh. 9 - Prob. 18QCh. 9 - Prob. 19QCh. 9 - Prob. 20QCh. 9 - Prob. 21QCh. 9 - Prob. 22QCh. 9 - Prob. 23QCh. 9 - Prob. 24QCh. 9 - Prob. 25QCh. 9 - Prob. 26QCh. 9 - Prob. 27QCh. 9 - Prob. 28QCh. 9 - Prob. 29QCh. 9 - Prob. 30QCh. 9 - Prob. 31QCh. 9 - Prob. 32QCh. 9 - Prob. 33QCh. 9 - Prob. 34QCh. 9 - Prob. 35QCh. 9 - Prob. 36QCh. 9 - Prob. 37QCh. 9 - Prob. 38QCh. 9 - Prob. 39QCh. 9 - Prob. 40QCh. 9 - Prob. 41QCh. 9 - Prob. 42QCh. 9 - Prob. 43QCh. 9 - Prob. 44QCh. 9 - Prob. 45QCh. 9 - Prob. 46QCh. 9 - Prob. 47QCh. 9 - Prob. 48QCh. 9 - Prob. 49QCh. 9 - Prob. 50QCh. 9 - Prob. 51QCh. 9 - Prob. 52QCh. 9 - Prob. 53QCh. 9 - Prob. 54QCh. 9 - Prob. 55QCh. 9 - Prob. 56QCh. 9 - Prob. 57QCh. 9 - Prob. 58QCh. 9 - Prob. 59Q
Additional Science Textbook Solutions
Find more solutions based on key concepts
The structural formula of 1, 2-dimethylbenzene needs to be drawn. Concept introduction: The ring structures of ...
Chemistry: Matter and Change
Q2. Which statement best defines chemistry?
a. The science that studies solvents, drugs, and insecticides
b. Th...
Introductory Chemistry (6th Edition)
22.102 Write the structures of the cis and tram isomers, if any, for the following compounds:
Chemistry: The Molecular Nature of Matter
Q1. What is the empirical formula of a compound with the molecular formula
Chemistry: A Molecular Approach
Consider a sample of ideal gas initially in a volume V at temperature T and pressure P. Does the entropy of thi...
General Chemistry: Principles and Modern Applications (11th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Structure A is identical or the same as Structure B. Compound B has a cis-structure. A O Both statements are true. Both statements are false. O Only the first statement is true. Only the second statement is true. Clear my choice Barrow_forwardWhat type(s) of intermolecular forces are expected between CH3CH,CH2CH2CH2CH3 molecules? H H H H H H- H- H. H H H H. Indicate with a Y (ves) or an N (no) which apply. dipole forces induced dipole forces hydrogen bonding Submit Answer Try Another Version 2 item attempts remainingarrow_forwardWhat type(s) of intermolecular forces are expected between CH3CH2CH2COOH molecules? H. H H H- OH H H Indicate with a Y (yes) or an N (no) which apply. dipole forces induced dipole forces hydrogen bonding Y Submit Answer Retry Entire Group 2 more group attempts remainingarrow_forward
- Rank the following molecules from highest to lowest boiling point temperatures Previous Page ethy methyl ether butanoic acid butanol methylpropane butane Next Page 1. Highest 2. 2nd highest 3. 3rd highest 4. 4th highest 5. Lowest Pagearrow_forwardNeed help only part Barrow_forwardorganic chemistry help?? 3 part questionarrow_forward
- Part 1. TRUE or FALSE.Choose A if the statement is true. Choose D if the statement is false. 1. Based on a technical definition, acids are classified as nucleophiles. 2. A nucleophile is any atom or molecule which can attack positively or partially positively charged molecules or atoms. 3. Covalent bonds are directional due to repulsion of orbitals. 4. All compounds which contain carbon are classified as organic compounds. 5. The density of an organic compound is influenced by the type of chemical bond present in the compound. 6. Vitalism proposes that living organisms formed from non-living things. 7. Urea, which has been successfully synthesized in the laboratory, has the chemical formula CO2N2H4. 8. The possible mathematical values of p-orbital are: p, pz and p. 9. The ability of carbon atoms to link with each other is termed catenation. 10. Pi bonds are occupied by a maximum of two electrons. 11. Organic chemistry studies carbon atom in the context of atomic properties. 12.…arrow_forwardes Mailings Aa A A 2 v Text Predictions On Review View Help Paragraph Accessibility Good to go 1 3 0 f4 101 f5 s f6 The difference between a covalent and an ionic bond is that the covalent always donates its electrons to create a polar and a nonpolar compound. Is it True or false ? An aldehyde is an example of a saturated organic compound? IF the 5 f7 mm Normal No Spacing Styles Heading 1 (Ctrl) - <<arrow_forwardjust need the HBrO4arrow_forward
- Which carbon(s) is/are electrophilic on the molecule shown below? 4 5 3 60 A. 1 B. 1 and 2 C. 1 and 3 D. 2 E. 2 and 3 F. 3arrow_forwardGood explanation asaparrow_forward1. Rank the following with increasing acidity: CH3OH, HCl, NH3, and CH4 2. Select the correct molecule with the lowest surface tension. a. CH3CH2CH2OH b. CH3CH2CH2NH2 c. CH3CH2CH2CH3 d. CH3CH2CH2Clarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Quantum Mechanics - Part 1: Crash Course Physics #43; Author: CrashCourse;https://www.youtube.com/watch?v=7kb1VT0J3DE;License: Standard YouTube License, CC-BY