
A gas turbine operates with a regenerator and two stages of reheating and intercooling. Air enters this engine at 14 psia and 60°F, the pressure ratio for each stage of compression is 3, the air temperature when entering a turbine is 940°F, the engine produces 1000 hp, and the regenerator operates perfectly. The isentropic efficiency of each compressor is 88 percent and that of each turbine is 93 percent. Which process of the cycle loses the greatest amount of work potential? The temperature of the heat source is the same as the maximum cycle temperature, and the temperature of the heat sink is the same as the minimum cycle temperature. Use constant specific heats at room temperature.
Which process of the cycle loses the greatest amount of work potential.
Answer to Problem 151P
The exergy destruction associated with process 1-2 and 3-4 is
The exergy destruction associated with process 5-6 and 7-8 is
The exergy destruction associated with process 6-7 and 8-9 is
The exergy destruction associated with process 10-1 and 2-3 is
The exergy destruction associated at regenerator is
During the heat rejection process the highest energy destruction occurs.
Explanation of Solution
Draw the
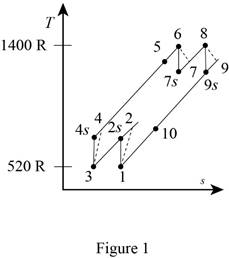
Write the expression for the temperature and pressure relation for the isentropic process 1-2s.
Here, the pressure ratio is
Write the expression for the efficiency of the compressor
Here, the specific heat at constant pressure is
Write the expression for the temperature and pressure relation ratio for the expansion process 6-7s.
Here, temperature at state 7s for isentropic process is
Write the expression for the efficiency of the turbine
Here, temperature at state 7 is
Write the expression to calculate the heat input for the two-stage gas turbine
Here, the specific heat of air at constant pressure is
Write the expression to calculate the heat output for the two-stage gas turbine
Write the expression for the exergy destruction during the process of as steam from an inlet to exit state.
Here, entropy generation is
Write the expression of exergy destruction for process 1-2
Here, pressure at state 2 is
Write the expression of exergy destruction for process 5-6
Here, pressure at state 5 is
Write the expression of exergy destruction for process 6-7
Here, pressure at state 7 is
Write the expression of exergy destruction for process 10-1
Here, pressure at state 10 is
Write the expression of exergy destruction for regenerator
Conclusion:
Substitute
Substitute
Substitute
Substitute
The regenerator is ideal, the effectiveness is 100% and therefore,
Substitute
Substitute
Substitute
Thus, the exergy destruction associated with process 1-2 and 3-4 is
Substitute
Thus, the exergy destruction associated with process 5-6 and 7-8 is
Substitute
Thus, the exergy destruction associated with process 6-7 and 8-9 is
Substitute
Thus, the exergy destruction associated with process 10-1 and 2-3 is
Substitute
Thus, the exergy destruction associated at regenerator is
During the heat rejection process the highest energy destruction occurs.
Want to see more full solutions like this?
Chapter 9 Solutions
THERMODYNAMICS (LL)-W/ACCESS >IP<
- Consider a two stage compression and two stage expansion in an ideal gas-turbine cycle. The air enters each stage of the compressor at 350 K and each stage of the turbine at 1240 K. The pressure ratio across each stage of the compressor and turbine is 3.5. Assuming an efficiency of 82 percent for each compressor stage and an efficiency of 88 percent for each turbine stage. Determine the thermal efficiency of the cycle, assuming (a) no regenerator is used and (b) a regenerator with 85 percent effectiveness is used.arrow_forwardAir enters the compressor of a gas turbine at 100 kPa and 25°C. Determine the back work rate and thermal efficiency of the Brayton cycle for a pressure ratio of 5 and a maximum temperature of 850°C.arrow_forwardAir enters the compressor of a regenerative gasturbine engine at 310 K and 100 kPa, where it is compressed to 900 kPa and 650 K. The regenerator has an effectiveness of 80 percent, and the air enters the turbine at 1400 K. For a turbine efficiency of 90 percent, determine the thermal efficiency. Assume variable specific heats for air.arrow_forward
- Answer All! Air enters the compressor of a gas turbine at 100 kPa and 300 K with a volume flow rate of 5 m3/s. The compressor pressure ratio is 10 and its isentropic efficiency is 85%. At the inlet to the turbine, the pressure is 950 kPa and the temperature is 1400 K. The turbine has an isentropic efficiency of 88% and the exit pressure is 100 kPa. On the basis of an air-standard analysis, what is the thermal efficiency of the cycle in percent?a. 42.06 c. 31.89b. b. 60.20 d. 25.15 in a gas turbine operating on the air-standard cycle, the air enters the compressor at 100 kPa and 30oC at the rate of 20 m3/s and is compressed to 500 kPa. The maximum temperature is 780oC and the exit pressure of the turbine is 100 kPa. Determine the net turbine power.a. 4853 kW c. 4483 kWb. 4358 kW d. 4538 kW A gas turbine working on air-standard Brayton cycle has air enter into the compressor at atmospheric condition and 22oC. The pressure ratio is 9 and the maximum temperature in…arrow_forwardAir in an ideal Diesel cycle is compressed from 2 to 0.13 L, and then it expands during the constant-pressure heataddition process to 0.30 L. Under cold-air-standard conditions, the thermal efficiency of this cycle is (a) 41 percent (b) 59 percent (c) 66 percent (d) 70 percent (e) 78 percentarrow_forwardHelium is used as the working fluid in a Brayton cycle with regeneration. The pressure ratio of the cycle is 8, the compressor inlet temperature is 300 K, and the turbine inlet temperature is 1800 K. The effectiveness of the regenerator is 75 percent. Determine the thermal efficiency and the required mass flow rate of helium for a net power output of 60 MW, assuming both the compressor and the turbine have an isentropic efficiency of (a) 100 percent and (b) 80 percent.arrow_forward
- Helium is used as the working fluid in a Brayton cycle with regeneration. The pressure ratio of the cycle is 8, the compressor inlet temperature is 300 K, and the turbine inlet temperature is 1800 K. The effectiveness of the regenerator is 75 percent. Determine the thermal efficiency and the required mass flow rate of helium for a net power output of 60 MW, assuming both the compressor and the turbine have an isentropic efficiency of 100 percent.arrow_forwardA four-cylinder, four-stroke, 1.6-L gasoline engine operates on the Otto cycle with a compression ratio of 11. The air is at 100 kPa and 37°C at the beginning of the compression process, and the maximum pressure in the cycle is 8 MPa. The compression and expansion processes may be modeled as polytropic with a polytropic constant of 1.3. Using constant specific heats at 850 K, determine the mean effective pressure.\arrow_forwardThe temperature of air at the beginning of compression in an Otto cycle is 310 K and maximum temperature in the cycle is 2200 K. If the pressure at the end of adiabatic compression is 15 times that at the start, determine the compression ratio, thermal efficiency, and work done. Take k for air = 1.4.arrow_forward
- In an ideal Brayton cycle, air is compressed from 100 kPa and 25°C to 1 MPa, and then heated to 927°C before entering the turbine. Under cold-air-standard conditions, the air temperature at the turbine exit is (a) 349°C (b) 426°C (c) 622°C (d) 733°C (e) 825°Carrow_forwardAs a car gets older, will its compression ratio change? How about the mean effective pressure?arrow_forwardIn an ideal Brayton cycle, air is compressed from 100 kPa and 25°C to 1 MPa, and then heated to 1000°C before entering the turbine. The air temperature at the turbine exit isarrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





