
(a)
The wt % proeutectic
(a)
Answer to Problem 92AAP
The wt % proeutectic
Explanation of Solution
Refer Figure 9.43, “Aluminum-rich end of aluminum-copper phase diagram”.
Draw the plot with the notation
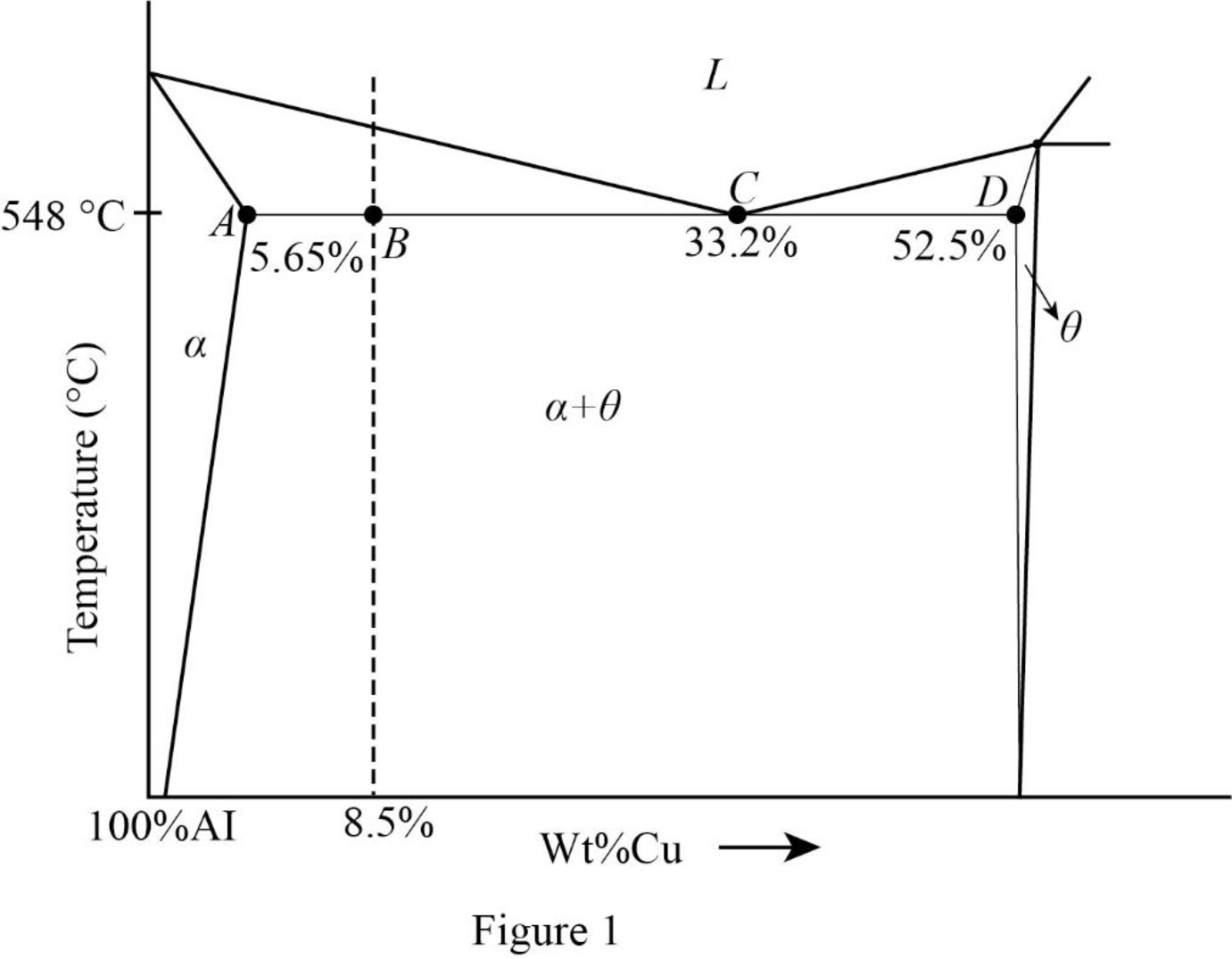
Refer Figure 1,
The weight percentage of proeutectic
Thus, the wt % proeutectic
(b)
The wt % eutectic
(b)
Answer to Problem 92AAP
The wt % eutectic
Explanation of Solution
Refer Figure 1,
The weight percentage of total
The weight percentage of eutectic
Thus, the wt % eutectic
(c)
The wt %
(c)
Answer to Problem 92AAP
The wt %
Explanation of Solution
Refer Figure 1,
The weight percentage of
Thus, the wt %
Want to see more full solutions like this?
Chapter 9 Solutions
Connect Access Card For Foundations Of Materials Science And Engineering
- For a 70-30 Zn-Cu alloy, what would the percent of each phase be at 800°C and 500°C?arrow_forwardYou are assured that a Pb-98.5%Sn alloy is good for a treatment of Aging, if we take that information as correct, will it be possible that an alloy Is Pb-10%Sn also very good for that treatment? Justify your answer.arrow_forwardFor a 60-40 Pb-Sn alloy, what would the percent of each phase be at 185°C and 180°C?arrow_forward
- When alloys A, B, and C are gently cooled from 1000 o C in the following temperature ranges, sketch their microstructures. (equilibrium cooling). List every phase present in the microstructures. Comment on the constituent phases of the resulting alloy and its mechanical characteristics. Please keep it brief.arrow_forwardA 80%Ag-20%Cu alloy is slowly heated from room temperature (25°C).Determine (1) At what temperature does the first liquid phase form and what is this liquid’s composition?(2) At what temperature does all solid melt and what is the last remaining solid’s composition?arrow_forwardBriefly describe how precipitation hardening happens in Al alloys (how the microstructure evolves as annealing time increases). Why it first forms q” then q’ before q is finally formed? How does the alloy’s strength change during this process? What are the strengthening mechanisms?arrow_forward
- For a 50-50 Mg-Pb alloy, what would the percent of each phase be at 500°C, 300°C and 100°C?arrow_forwardfind the relative amounts (in terms of mass fractions) of the phases for the following alloys and temperature: 90 wt% Zn-10 wt% Cu at 400°C.arrow_forwardAn alloy of 30 wt % Pb and 70 wt % Sn is slowly cooled from 250°C to 27°C: a) Is this alloy hypoeutectic or hypereutectic? b) What is the composition of the first solid to form? c) What are the amounts and compositions of each phase that is present at 183°C + ∆T ? d) What is the amount and composition of each phase that is present at 183°C - ∆T ? e) What are the amounts of each phase present at room temperature? f) Draw the microstructure 183°C + ∆T and 183°C - ∆T in 2 cm diameter circle.arrow_forward
- The following questions are based on a high carbon steel with a composition of 1,5% C and 98,5% Fe. Please refer to the Fe-Fe3C binary phase diagram in Callister and Rethwhich 1) a Name the phases present and calculate the amounts of these phases just above the eutectoid temperature b, Give the amount of austenite present at 1100°C. c. Give the amount of pro-eutectoid ferrite just above the eutectoid temperature. d. With equilibrium cooling just below the eutectoid temperature calculate the total ferrite. e. Why must the samples be quenched rapidly to obtain high hardness?arrow_forwardDraw Fe-C binary phase diagram and discuss martensitic transformation for 0.1C% and 0.5C% plain carbon steels.arrow_forwardA 50 wt% Pb–50 wt% Mg alloy is slowly cooled from 700°C to 400°C. (a) At what temperature does the first solid phase form? (b) What is the composition of this solid phase? (c) At what temperature does the liquid solidify? (d) What is the composition of this last remaining liquid phase?arrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





