
EBK ORGANIC CHEMISTRY AS A SECOND LANGU
4th Edition
ISBN: 9781119234722
Author: Klein
Publisher: JOHN WILEY+SONS,INC.-CONSIGNMENT
expand_more
expand_more
format_list_bulleted
Question
Chapter 9.3, Problem 9.19P
Interpretation Introduction
Interpretation:
Efficient synthesis for the given transformation has to be suggested.

Concept Introduction:
Ketone or
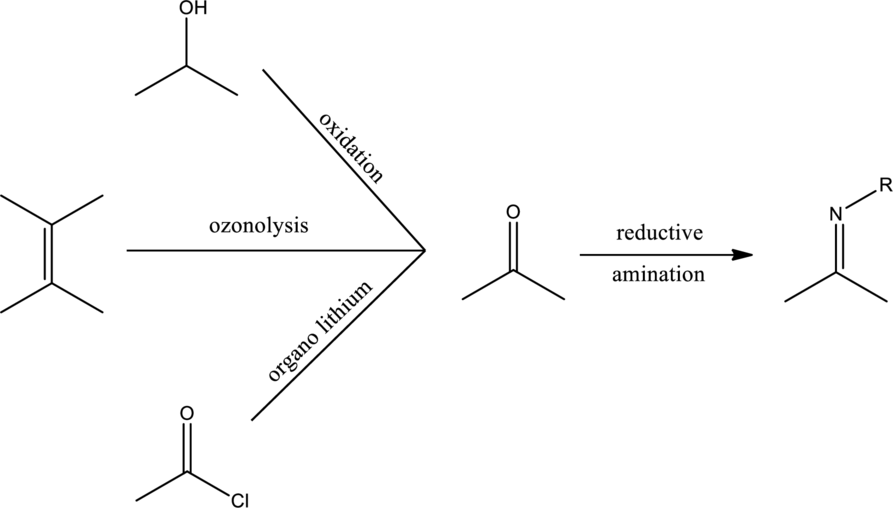
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Propose syntheses for the product shown on the left starting from the indicated compound (shown on the right). Your answer should include a retrosynthetic analysis and a proposed synthetic route with reagents.
Benzyl acetate is a pleasant-smelling ester found in the essential oil of jasmine flowers and is used in many perfume formulations.
Starting with benzene and using any other reagents of your choice, design an efficient synthesis for benzyl acetate.
The transformation above can be performed with some reagent or combination of the reagents listed below. Give the necessary
reagents in the correct order, as a string of letters (without spaces or punctuation, such as "EBF"). If there is more than one correct
solution, provide just one answer.
A. 1)
D.
i
; 2) H3O
+ AICI 3
CI
G. NBS, heat
B. 1)
요.
'H
H
; 2) H3O+
Ei
E.
H. 1) LiAlH4; 2) H3O+
CI
+ pyridine
C. 1)
F. Mg
i
I. Br2, AlBr3
2) H3O+
A girl must convert 1-butyne into 2-pentanione. Assist her by recommending a synthetic plan to follow in order to obtain the desired result. Indicate all of the reagents she'll require.
Chapter 9 Solutions
EBK ORGANIC CHEMISTRY AS A SECOND LANGU
Ch. 9.2 - Identify how you would use a Gabriel synthesis to...Ch. 9.2 - Identify how you would use a Gabriel synthesis to...Ch. 9.2 - Identify how you would use a Gabriel synthesis to...Ch. 9.2 - Identify how you would use a Gabriel synthesis to...Ch. 9.2 - Identify whether each of the following compounds...Ch. 9.2 - Identify whether each of the following compounds...Ch. 9.2 - Identify whether each of the following compounds...Ch. 9.2 - Identify whether each of the following compounds...Ch. 9.3 - Suggest an efficient synthesis for each of the...Ch. 9.3 - Suggest an efficient synthesis for each of the...
Ch. 9.3 - Suggest an efficient synthesis for each of the...Ch. 9.3 - Suggest an efficient synthesis for each of the...Ch. 9.3 - Suggest an efficient synthesis for each of the...Ch. 9.3 - Prob. 9.18PCh. 9.3 - Prob. 9.19PCh. 9.3 - Prob. 9.20PCh. 9.3 - Prob. 9.21PCh. 9.3 - Prob. 9.22PCh. 9.4 - Prob. 9.24PCh. 9.4 - Prob. 9.25PCh. 9.5 - Predict the major product for each of the...Ch. 9.5 - Predict the major product for each of the...Ch. 9.5 - Predict the major product for each of the...Ch. 9.5 - Predict the major product for each of the...Ch. 9.6 - What reagents would you use to achieve each of the...Ch. 9.6 - What reagents would you use to achieve each of the...Ch. 9.6 - What reagents would you use to achieve each of the...Ch. 9.6 - What reagents would you use to achieve each of the...Ch. 9.6 - What reagents would you use to achieve each of the...
Knowledge Booster
Similar questions
- Propose a synthetic route to the following molecule. Use any saturated ketone, aldehyde, or ester you need (ie: you can’t use any dicarbonyl or a,b-unsaturated carbonyl compounds. You have to make those). Please show a reaction sequence.arrow_forwardPropose a synthesis for the following product from the starting material(s) indicated. Show all reagents and products of each step of your sequence. Show retrosynthetic analysis. фон OH OHarrow_forwardPropose a synthetic route to convert the starting material shown below into the indicated target molecule. You may also use any stable molecule with three or fewer carbons and any commercially available reagents. You must show how to generate any organometallic reagents you use. Please show all reagents and intermediates in your synthesis. You do not need to show a reaction mechanism. OH youarrow_forward
- Benzyl acetate is a pleasant-smelling ester found in the essential oil of jasmine flowers and is used in many perfume formulations. Starting with benzene and using any other reagents of your choice, design an efficient synthesis for benzyl acetate. o-one The transformation above can be performed with some reagent or combination of the reagents listed below. Give the necessary reagents in the correct order, as a string of letters (without spaces or punctuation, such as "EBF"). If there is more than one correct solution, provide just one answer. A. 1) D. i ; 2) H₂O CI G. NBS, heat + AICI 3 B. 1) E. H eTextbook and Media i 'H ; 2) H3O+ + pyridine CI H. 1) LIAIH4; 2) H3O+ C. 1) 요 F. Mg 1. Br2, AlBr3 2) H3O+arrow_forwardShow how to bring about the following transformation.arrow_forwardPropose a synthesis for the given compound.arrow_forward
- Provide a synthesis for the target molecule shown below, starting with compounds that contain no more than two carbon atoms. Show your retrosynthetic analysis, and then provide a complete synthesis, showing all necessary reagents.arrow_forwardPropose the best synthesis for each of the following molecules. All of the carbons in your target molecule must come from the organic molecules attached to this question. Thank you.arrow_forwardShow how you could achieve the following transformation. Be sure to show the product produced after each reaction. You do not need to show mechanisms or a retrosynthesisarrow_forward
- When 2,2-dimethylcyclohexanol reacts with acid in water, multiple products form. One of these products is shown below. Provide a complete mechanism for the following transformation using only the reagents listed in the reaction scheme below.Show the intermediate(s) in the blank box(es) and draw all electrons and charges. In each box, only show one mechanistic transformation. Include only the reagents that are reacting at the time; do not show any byproducts.arrow_forwardDraw a possible forward synthesis to form the following compounds using MeOH, ETOH, NaCN and benzene as the only carbon sources. You can use any other reagents so long as they are not a source of carbon in the target molecule. A synthesis may use any previous compound as a reagent. Use retrosynthetic analysis to help. There may be more than one correct answer to each problem. НО Но OH j) k) h) HNarrow_forwardHi! Could someone help me with the following question, any help is appreciated! Provide complete mechanisms for the following transformations.Include arrow notations, resonance forms, and intermediates. Please include reagents. Form the following from ethyne.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY