
Concept explainers
(a)
Interpretation:
The major product expected when given
Concept Introduction:
Addition Reaction: It is defined as
Markovnikov’s rule: An unsymmetrical

Anti-Markovnikov’s rule: An unsymmetrical alkene reacts with hydrogen halide in which halide ions goes to the less substitution position of carbon-carbon double bond which provides alkyl halides.
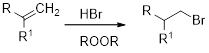
(b)
Interpretation:
The major product expected when given alkyne reacts with aqueous acid has to be drawn.
Concept Introduction:
Addition Reaction: It is defined as chemical reaction in which two given molecules combines and forms product. The types of addition reactions are electrophilic addition, nucleophilic addition, free radical additions and cycloadditions. Generally, compounds with carbon-hetero atom bonds favours addition reaction.
Markovnikov’s rule: An unsymmetrical alkene reacts with hydrogen halide in which halide ions goes to the more substitution position of carbon-carbon double bond which provides alkyl halides.

Anti-Markovnikov’s rule: An unsymmetrical alkene reacts with hydrogen halide in which halide ions goes to the less substitution position of carbon-carbon double bond which provides alkyl halides.
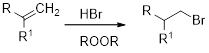
(c)
Interpretation:
The major product expected when given alkyne reacts with aqueous acid has to be drawn.
Concept Introduction:
Addition Reaction: It is defined as chemical reaction in which two given molecules combines and forms product. The types of addition reactions are electrophilic addition, nucleophilic addition, free radical additions and cycloadditions. Generally, compounds with carbon-hetero atom bonds favours addition reaction.
Markovnikov’s rule: An unsymmetrical alkene reacts with hydrogen halide in which halide ions goes to the more substitution position of carbon-carbon double bond which provides alkyl halides.

Anti-Markovnikov’s rule: An unsymmetrical alkene reacts with hydrogen halide in which halide ions goes to the less substitution position of carbon-carbon double bond which provides alkyl halides.
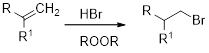
(d)
Interpretation:
The major product expected when given alkyne reacts with aqueous acid has to be drawn.
Concept Introduction:
Addition Reaction: It is defined as chemical reaction in which two given molecules combines and forms product. The types of addition reactions are electrophilic addition, nucleophilic addition, free radical additions and cycloadditions. Generally, compounds with carbon-hetero atom bonds favours addition reaction.
Markovnikov’s rule: An unsymmetrical alkene reacts with hydrogen halide in which halide ions goes to the more substitution position of carbon-carbon double bond which provides alkyl halides.

Anti-Markovnikov’s rule: An unsymmetrical alkene reacts with hydrogen halide in which halide ions goes to the less substitution position of carbon-carbon double bond which provides alkyl halides.
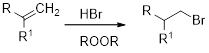
Want to see the full answer?
Check out a sample textbook solution
Chapter 9 Solutions
ORGANIC CHEMISTRY (LL) >CUSTOM PACKAGE<
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





