
a)
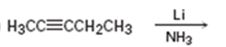
Interpretation:
The product formed and the electron pushing mechanism for its formation, when 2-pentyne is reduced with Li in NH3 is to be given.
Concept introduction:
Lithium metal donates an electron to the
The addition takes place with trans stereochemistry. The stereochemistry is established in the third step when the less hindered vinylic anion is formed from vinylic radical.
To propose:
The product formed and the electron pushing mechanism for its formation, when 2-pentyne is reduced with Li in NH3.
b)
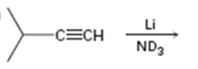
Interpretation:
The product formed and the electron pushing mechanism for its formation, when 3-methyl-1-butyne is reduced with Li in ND3 is to be given.
Concept introduction:
Lithium metal donates an electron to the alkyne to give an anion radical which in the next step abstracts a proton from deuteratedammonia solvent to yield a vinylic radical. In the third step the vinylic radical accepts a second electron from another Li atom to produce a vinylic anion which in the fourth step abstracts another proton from the solvent to yield the final trans product.
The addition takes place with trans stereochemistry. The stereochemistry is established in the third step when the less hindered vinylic anion is formed from vinylic radical.
To propose:
The product formed and the electron pushing mechanism for its formation, when 3-methyl-1-butyne is reduced with Li in ND3.
c)
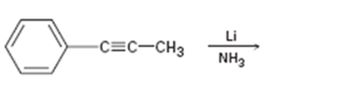
Interpretation:
The product formed and the electron pushing mechanism for its formation, when propynylbenzene is reduced with Li in NH3 is to be given.
Concept introduction:
Lithium metal donates an electron to the alkyne to give an anion radical which in the next step abstracts a proton from ammonia solvent to yield a vinylic radical. In the third step the vinylic radical accepts a second electron from another Li atom to produce a vinylic anion which in the fourth step abstracts another proton from ammonia solvent to yield the final trans product.
The addition takes place with trans stereochemistry. The stereochemistry is established in the third step when the less hindered vinylic anion is formed from vinylic radical.
To propose:
The product formed and the electron pushing mechanism for its formation, when propynylbenzene is reduced with Li in NH3.
Trending nowThis is a popular solution!

Chapter 9 Solutions
Organic Chemistry (Instructor's)
- Give the complete detailed mechanism for the following reactionarrow_forwardClassify the following transformation as oxidation, reduction, or neither. Select the single best answer. oxidation reduction neither Give detailed Solution with explanation needed of all options. don't give Handwritten answerarrow_forwardprovide the product and mechanism for the following reactionarrow_forward
- Give the major organic products for each of the following reactions.arrow_forwardPredict the products of the following reduction reactions, including stereochemistry where needed. If the reaction product is racemic, indicate that by writing “racemic”. please explain stepsarrow_forwardSHOWS THE COMPLETE REACTION MECHANISM FOR THE FOLLOWING REDUCTION:arrow_forward
- Give the major elimination product for the following reactionarrow_forwardPlease give detailed mechanism for this reaction. Thank youarrow_forwardFor each of the following, show the mechanism(s), write the MAJOR product(s), and, whenapplicable, show or write the stereochemistry of the product(s).arrow_forward
- Complete the reaction scheme shown belowarrow_forward(a) A student was given a task to synthesize the products as shown below. Analyze the reaction schemes below and state whether the reaction conditions supplied in each reaction scheme will result in the products shown as MAJOR PRODUCTS. If wrong products are shown, draw the correct products and provide mechanistic details (curly arrows) of how the right (major) products were formed. E CI t-BuOK t-BuOHarrow_forwardGive the major products for the following reactionarrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


