
ORGANIC CHEMISTRY-WILEYPLUS ACCESS PKG.
12th Edition
ISBN: 9781119766919
Author: Solomons
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter D, Problem 10PP
Practice Problem D.10
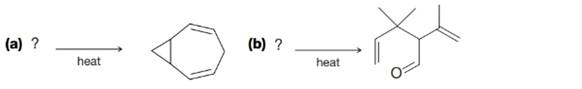
What reactant could lead to each product by either a Cope or Claisen rearrangement?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Problem 2,8
Suggest a plausible arrow-pushing mechanism for the following tautomerization
reactions
cat. HA
OH
cat B
O NH2
NH2
NIH
PROBLEM 3
Suggest mechanisms for these eliminations. Why does the first give a mixture
and the second a single product?
OH
OH
H₂PO4
heat
X³
OH
48% HBr
heat
64% yield, 4:1 ratio
OChem Question:
Starting with propyne and using any other required reagents, show how you would synthesize
(2R, 3R)- and (2S, 3S)-2,3-dibromobutane (as a racemic form)
Chapter D Solutions
ORGANIC CHEMISTRY-WILEYPLUS ACCESS PKG.
Ch. D - Prob. 1PPCh. D - Practice Problem D.2 (a) Show the orbitals...Ch. D - Prob. 3PPCh. D - Prob. 4PPCh. D - Prob. 5PPCh. D - Prob. 6PPCh. D - Practice Problem D.7 Can you suggest a...Ch. D - Practice Problem D.8 When compound A is heated,...Ch. D - Prob. 9PPCh. D - Practice Problem D.10 What reactant could lead to...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Q1. A chemist mixes sodium with water and witnesses a violent reaction between the metal and water. This is bes...
Chemistry: A Molecular Approach (4th Edition)
Propose a mechanism for each of the following transformations:
Organic Chemistry As a Second Language: Second Semester Topics
The heat capacity at constant pressure of a gas is determined experimentally at several temperatures, with the ...
Elementary Principles of Chemical Processes, Binder Ready Version
6.1 State the number of electrons that be must be lost by atoms of each of the following to achieve a stable el...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
What distinguishes the mass spectrum of 2,2-dimethylpropane from the mass spectra of pentane and isopentane?
Organic Chemistry
Determine the number of protons, neutrons, and electrons in the following atoms: a. a hydrogen atom that has a ...
General, Organic, and Biological Chemistry (3rd Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Problem 3 of 16 Draw the products of this reduction of a ketone with sodium borohydride. 1) NaBH4 49 Use a dash or wedge bond to indicate the stereochemistry of substituents on asymmetric centers, where applicableIgnore any inorganic byproducts. 2) HCI/H₂O Submit Select to Drawarrow_forwardProblem 28 of 50 Ignore stereochemistry. Q & Br dissociati on NaBr I Select to Draw I Carbocation Intermediate I I Submit I Carbocati on Rearral ngement Select to Draw I Carbocation Intermediatearrow_forwardProblem 3. Propose the synthesis of chalcone A from the two provided building blocks as the sources of carbon and any other reagents that needed. Br + H CF3 A CF3arrow_forward
- When both carbons ortho to the aryl oxygen are not bonded to hydrogen,an allyl aryl ether rearranges to a para-substituted phenol. Draw astepwise mechanism for the following reaction, which contains two [3,3]sigmatropic rearrangements.arrow_forwardProblem 27 of 50 Submit Curved arrows are used to illustrate the flow of electrons. Follow the curved arrows and draw the missing reactants/ intermediates in this SN1 mechanism. Include all lone pairs and charges as appropriate. Ignore byproducts. Ignore stereochemistry. Select to Draw Alkyl Halide dissociati on CH 02 H H 1,2- hydride shiftarrow_forwardProblem 29 of 50 Submit Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic steps. Be sure to account for all bond- breaking and bond-making steps. N Select to Add Arrows THF Na Ⓒ Na O Oarrow_forward
- Mechanism preparation of Aniline from nitrobenzenearrow_forwardProblem 17.9: Predict the major products in each of the following. Please show the complete CO CN mechanisms, Thanks 1. LIAIH,, ether 2. Но (a) H,C. 1. LIAIH, ether 2. H,00 (b) 10arrow_forwardQuestion 15 What synthetic goal is achieved by subjecting an alkene to a hydroboration-oxidation sequence? O syn-dihydroxylation anti-Markovnikov addition of H2O with anti stereochemistry O Markovnikov addition of H2O wherein skeletal rearrangement is promoted O Markovnikov addition of H2O wherein skeletal rearrangement is prevented anti-Markovnikov addition of H2O with syn stereochemistryarrow_forward
- Problem 12.17: Draw the mechanism for the reaction below. MCPBA Problem 12.18: What alkene can be epoxidized using MCPBA to yield the following compound?arrow_forwardProblem 15.3b Homework Predict the major organic products of the following reaction and draw their structures in the space provided. If appropriate, assume an aqueous workup at the end of the reaction. O HO + NaOMe 7 Open in Reading Viarrow_forwardProblem 11 of 32 Draw the product of the substitution reaction shown below. Ignore the inorganic byproducts. CH3CH₂I DMSO OO (+) Submit Naarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Enzymes - Effect of cofactors on enzyme; Author: Tutorials Point (India) Ltd;https://www.youtube.com/watch?v=AkAbIwxyUs4;License: Standard YouTube License, CC-BY
Enzyme Catalysis Part-I; Author: NPTEL-NOC IITM;https://www.youtube.com/watch?v=aZE740JWZuQ;License: Standard Youtube License