
Concept explainers
Interpretation:
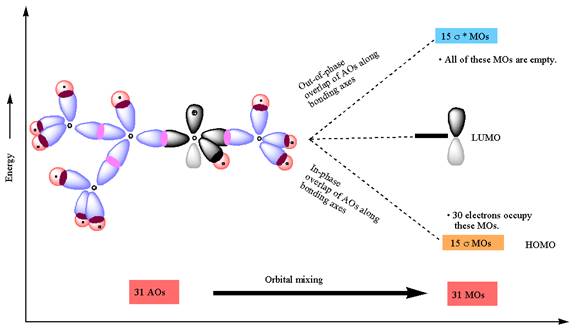
The atomic orbital overlap and MO energy diagrams for
Concept introduction:
According to
The molecular orbitals are formed by overlapping of atomic orbitals of adjacent atoms. The numbers of molecular orbitals formed are equal to the number of atomic orbitals overlap. The two atomic orbitals, on mixing along bonding axes, form two molecular orbitals, one is
Answer to Problem D.15YT
The atomic orbital overlap and MO energy diagrams for
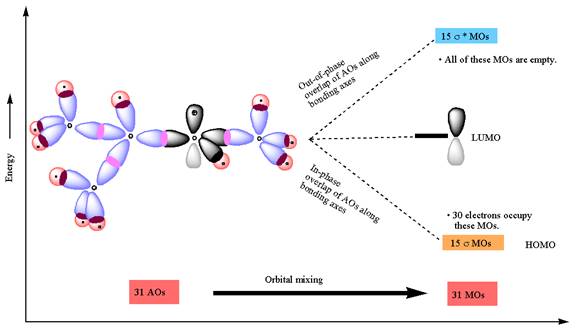
The constructed MO energy diagrams for
Explanation of Solution
In
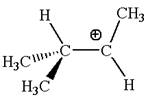
In forming the
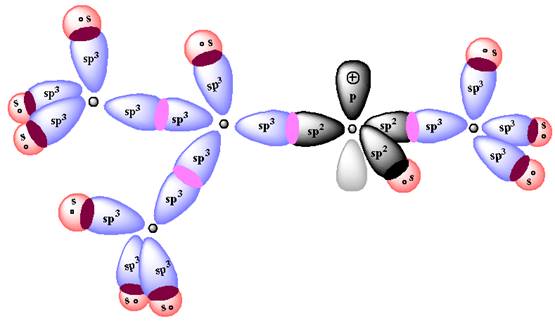
According to Aufbau Principle, the
The energy diagram for the formation of
The Figure

The constructed MO energy diagrams for
The energy diagram for the formation of
Want to see more full solutions like this?
Chapter D Solutions
EBK GET READY FOR ORGANIC CHEMISTRY
- A type of hyperconjugation occurs in the F-CH2-NH¯ anion even though there are no double bonds, triple bonds, or empty p AOs. Draw the orbital interaction that illustrates the most significant hyperconjugation interaction in this species. Hint: What is the HOMO in this species? Which adjacent o* orbital is the lowest in energy?arrow_forwardPlease don't provide hand writtin sollution...arrow_forwardPlace the following in order of increasing dipole moment. |- HI Il - HF III - HCI |< || = |II Il < III < | | < || < |II O Il < I < II OI< III < |arrow_forward
- 5. Molecular Orbitals 2. The carbon-oxygen single bond of carbonyls can be constructed using a mix of both hybridization and MO theory. Construct a MO diagram of the o bond of a carbonyl by mixing an oxygen sp² orbital and a carbon sp² orbital. Make sure your diagram is complete with all necessary orbitals (drawings of them), orbital notations, the proper number of electrons, and an appropriate energy ranking. H3Carrow_forwardCan you please help with the organic chemistry question attached?arrow_forwardClick to see additional instructions Shown is a blank MO diagram depicting ONLY the valence shells. Fill in the electrons for the compound PS. How many electrons are in the T*3p orbitals? How many electrons are in the 03s orbital? What is the bond order for PS?arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
